Chugai News Releases are issued to provide stakeholders with the most up-to-date information related to our company. In some instances, information on products or drug candidates under development may be included, but this is intended for members of the media, shareholders, and investors. The information is not intended for promotional or advertising purposes, or as medical advice, etc.
Apr 23, 2020
Chugai Announces 2020 1st Quarter Results
- Record-high revenues and core operating profit for the first quarter at ¥179.4 billion (+16.3%) and ¥74.1 billion (+54.7%), respectively
- Double-digit increases in both revenues and profits mainly driven by the contribution of overseas sales, and royalties and other operating income
- Good progress in research and development including the initiation of a clinical study for the first Switch Antibody project STA551
TOKYO, April 23, 2020 -- Chugai Pharmaceutical Co., Ltd. (TOKYO: 4519) announced its financial results for the first quarter of fiscal year 2020.
“We had a strong start to the year with record-high first quarter results driven by increased income related to our in-house product Hemlibra®. In terms of research and development, I am very pleased that we have initiated a clinical study for the first antibody project which applies our proprietary Switch Antibody engineering technology, and have also filed for regulatory approval of the liquid biopsy-based genomic profiling test FoundationOne® Liquid CDx. Going forward, the domestic market will be affected by drug price revisions, and we anticipate that the spread of the novel coronavirus will have an impact on business and performance in Japan and overseas. Nevertheless, we will continue striving to help realize advanced and sustainable patient-centric healthcare,” said Tatsuro Kosaka, Chugai’s Chairman and CEO.
[First quarter results for 2020]
Chugai reported record high revenues and operating profit for the first quarter (Core-basis), both achieving double-digit growth year-on-year.
Revenues increased by 16.3% due to increases both in sales and royalty and other operating income. Among sales, domestic sales increased by 2.6% driven by the contribution of mainstay products including the immune checkpoint inhibitor Tecentriq® and the hemophilia A treatment Hemlibra. Overseas sales increased by 10.9% mainly due to the commencement of export of Hemlibra to Roche at a regular shipment price. Royalties and other operating income increased by 110.2% due to a large increase in royalties and profit-sharing income for Hemlibra as well as an increase in other operating income with one-time income.
Operating profit increased by 54.7% due to the strong increase in royalties and profit-sharing income, and a better cost to sales ratio. Cost to sales ratio improved by 4.1 percentage point at 42.2% mainly due a larger proportion of in-house products in the total product mix.
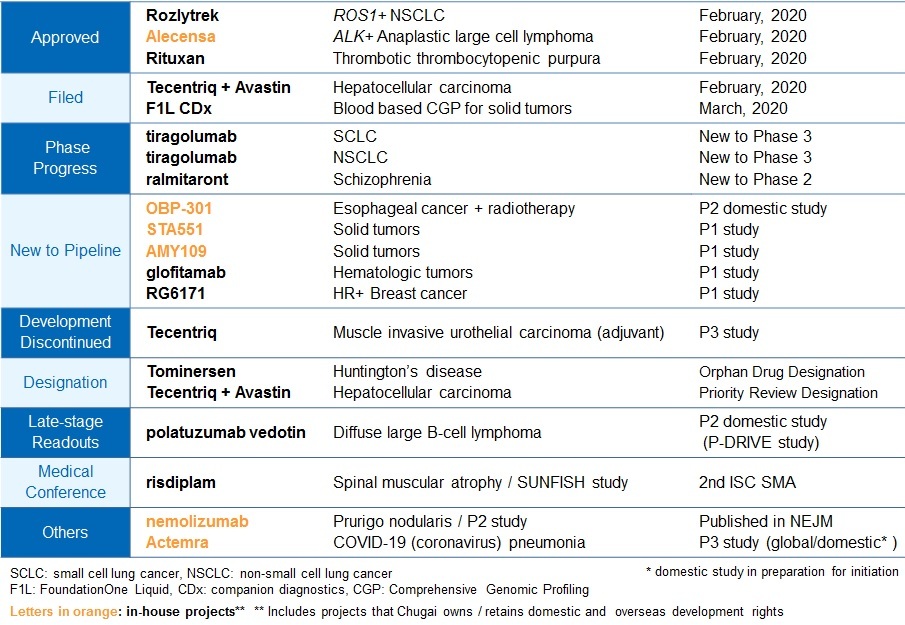
The Company also made good progress in research and development. Chugai started phase I study for STA551 for the treatment of solid tumors. STA551 is the first Switch Antibody project applying Chugai's proprietary antibody engineering technology. The regulatory application for FoundationOne Liquid CDx, a liquid biopsy-based genomic profiling test, was submitted in March for further contribution to cancer genomic medicine. Other projects also went well. A phase II study started in Japan for the oncolytic type 5 adenovirus OBP-301(telomelysin), an oncolytic virus therapy from Oncolys BioPharma Inc., in esophageal cancer. For the engineered antibody AMY109 from Chugai research, the Company started a phase I study in solid tumors.
[Impact on business and performance due to the spread of new coronavirus infection]
Chugai's response to the new coronavirus is to set up an Emergency Response Headquarters to take measures to prevent infection of employees and related business personnel, and to build a business continuity system centered on maintaining the product supply system. At present, there are no concerns about the product supply. During the three months under review, the impact of the new coronavirus on performance was minor. On the other hand, the Company anticipates that the impact on its business will be extensive from April, when the situation has worsened. Regarding the specific impact on business and performance, Chugai is currently collecting information from domestic and overseas.
[2020 first quarter results]
| Billion JPY | 2020 Jan - Mar | 2019 Jan - Mar | % change |
|---|---|---|---|
| Core results | |||
| Revenues | 179.4 | 154.3 | +16.3% |
| Sales | 144.5 | 137.7 | +4.9% |
| Royalties and other operating income | 34.9 | 16.6 | +110.2% |
| Operating profit | 74.1 | 47.9 | +54.7% |
| Net income | 52.7 | 36.3 | +45.2% |
| IFRS results | |||
| Revenues | 179.4 | 154.3 | +16.3% |
| Operating profit | 72.4 | 46.1 | +57.0% |
| Net income | 51.5 | 35.0 | +47.1% |
[Sales breakdown]
| Billion JPY | 2020 Jan - Mar | 2019 Jan - Mar | % change |
|---|---|---|---|
| Sales | 144.5 | 137.7 | +4.9% |
| Domestic sales | 101.9 | 99.3 | +2.6% |
| Oncology | 54.7 | 52.0 | +5.2% |
| Bone and joint diseases | 24.9 | 24.2 | +2.9% |
| Renal diseases | 6.7 | 7.9 | -15.2% |
| Others | 15.5 | 15.2 | +2.0% |
| Overseas sales | 42.6 | 38.4 | +10.9% |
[Progress in R&D activities from Jan 31st, 2020 to Apr 23rd, 2020]
About Core results
Chugai discloses its results on a Core basis from 2013 in conjunction with its decision to apply IFRS. Core results are the results after adjusting non-Core items to IFRS results, and are consistent with the Core concept disclosed by Roche. Core results are used by Chugai as an internal performance indicator, for explaining the underlying business performance both internally and externally, and the basis for payment-by-results such as a return to shareholders.
Trademarks used or mentioned in this release are protected by law.
Contact:
- For Media
- Chugai Pharmaceutical Co., Ltd.
- Media Relations Group, Corporate Communications Dept.,
- Tomoko Shimizu
- Tel: +81-3-3273-0881
- E-mail: pr@chugai-pharm.co.jp
- For Investors
- Chugai Pharmaceutical Co., Ltd.
- Investor Relations Group, Corporate Communications Dept.,
- Toshiya Sasai
- Tel: +81-3-3273-0554
- E-mail: ir@chugai-pharm.co.jp





