Creation of a System for Comprehensive Assessment of Suppliers
Backdrop of changes in the assessment system
Cooperation with suppliers is becoming imperative for companies in their efforts to solve social issues such as increasing poverty and inequality, environmental problems, and deteriorating labor conditions.
In this context, Chugai reexamined how it should cooperate with suppliers to respond to the changes and needs of society, and is working to build a system for comprehensive assessment of suppliers.
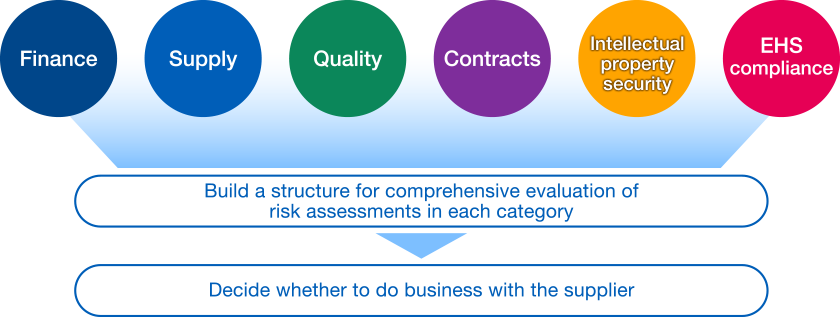
We have added the categories of environment, health and safety (EHS) and compliance (including corporate ethics and human rights) to our assessment criteria for suppliers in addition to the five categories that we previously assessed – financial condition, supply, quality, contracts, and intellectual property and security – to establish a more comprehensive assessment system. These additions reflect the fact that Chugai, as a pharmaceutical company, engages in many scientific and specialized activities in which it deals with materials that require handling precautions because of their potential effects on the human body; that the scope of environmental protection and health and safety management extends across the value chain; and that human rights problems such as child labor and forced labor are becoming serious global issues.
EHS and Compliance Risk Evaluation
We have established the Chugai Group Supplier Code of Conduct as a set of behavioral standards we expect of suppliers (including subcontractors) to do business with us, and we request that they comply with that code of conduct.
Based on the “Pharmaceutical Industry Principles for Responsible Supply Chain Management.” issued by the Pharmaceutical Supply Chain Initiative (PSCI), an organization made up of global pharmaceutical companies, the Chugai Group Supplier Code of Conduct delineates the items we want supplier to comply with in the areas of ethics, human rights, health and safety, environment and related governance & management systems.
Chugai Group Supplier Code of Conduct (v2.0; July 2025 revision)
PSCI (Pharmaceutical Supply Chain Initiative)
https://pscinitiative.org/home
- * Please visit PSCI website for details of “Pharmaceutical Industry Principles for Responsible Supply Chain Management.”
In addition, we have established the Guideline for EHS and Compliance Risk Evaluation of Suppliers and categorize our suppliers by analyzing risks to workers, local residents, and the surrounding environment using chemical substances and biological samples that are indispensable for pharmaceutical manufacturing, and conduct the following assessments according to the risks of suppliers.
- Assess the supplier’s reputation online
- Assess the results of supplier self-assessments of compliance with the Supplier Code of Conduct
- For major suppliers, conduct the on-site inspection of supplier compliance with the Supplier Code of Conduct, including employee interviews
- Settle on Corrective Action Plan and monitor them
We re-evaluate EHS and Compliance Risks every three years.
Furthermore, supplier feedback is a component of our practice of EHS/compliance evaluation. After our assessment to suppliers, we provide, as needed, the supplier with feedback that will be constructive to the responsible supply chain in terms of ethics, human rights, EHS and all the governance & management system for them.
The result of supplier*1 EHS/compliance risk evaluation
| 2023 | 2024 | |
|---|---|---|
| Number of suppliers which have agreed with SCC | 131 | 221 |
| Number of suppliers evaluated for EHS/Compliance risks | 28 | 34 |
| - Number of suppliers evaluated by SAQ | 19 | 20 |
| - Number of suppliers evaluated by SAQ and site audit | 9 | 14 |
| Number of Suppliers discontinued based on audit results | 0 | 0 |
- *1: Including contractors, e.g. Contract Manufacturing Organization(CMO) etc.
The findings issues and corrective actions of supplier*2 EHS/compliance risk evaluation
| Assessment Issues | Findings 2023 |
Corrective actions in place*4 | Corrective actions Completed | Corrective actions In progress |
|---|---|---|---|---|
| Ethics | 0 | 100% | 100% | 0% |
| Human Rights and Labor | 1(1)*3 | 100% | 100% | 0% |
| Safety and Health | 57(43)*3 | 100% | 14% | 86% |
| Environment | 34(29)*3 | 100% | 0% | 100% |
| Management System | 5(4)*3 | 100% | 0% | 100% |
| Assessment Issues | Findings 2024 |
Corrective actions in place*4 | Corrective actions Completed | Corrective actions In progress |
|---|---|---|---|---|
| Ethics | 3(3)*3 | 100% | 100% | 0% |
| Human Rights and Labor | 3(3)*3 | 100% | 100% | 0% |
| Safety and Health | 99(70)*3 | 100% | 97% | 3% |
| Environment | 57(34)*3 | 100% | 100% | 0% |
| Management System | 13(10)*3 | 100% | 33% | 67% |
- *2: The supplier here is the CMO.
- *3: Number of findings in parentheses came from due diligence activities before contract.
- *4: These corrective actions are in progress.
Cooperation with PSCI
Chugai became a member of the PSCI in November 2018.
PSCI is building a platform for member companies to cooperate in conducting supplier assessments. For a pharmaceutical company, assessing the EHS and compliance risks of suppliers on its own is costly and time-consuming. Therefore, the PSCI is setting up a framework that will enable member companies to use each other’s evaluation results. This framework will allow assessments to be conducted in a way that is efficient and effective for both pharmaceutical companies and suppliers.
EHS and Compliance Risk Evaluation Results and Future Targets
We have focused on implementing EHS and Compliance Risk Evaluation with contract manufacturers as “major suppliers” due to the impact of EHS and Compliance risks and the degree of difficulty in selecting alternative suppliers. Evaluation of all existing contract manufacturers was completed by the end of 2022. Even after the evaluation, they will be monitored continuously thereafter. In addition, we plan to extend the scope of our evaluation to include the secondary suppliers by 2030.