
What kind of company is Chugai?
Chugai’s features and strengths are presented in six Q&As.
Q1What kind of drugs does Chugai deal with?
We specialize in prescription drugs and deliver innovative pharmaceuticals to patients around the world.
Prescription
drugs are drugs prescribed by a physician or administered based on
a physician’s prescription. Therefore, these products are not seen
on store shelves.
In 2004, Chugai divested its
over-the-counter (OTC) drug business, which included energy drinks
and insecticides. Since then, we have focused on the prescription
drugs business, which includes the research, development,
manufacturing, and marketing of prescription drugs.
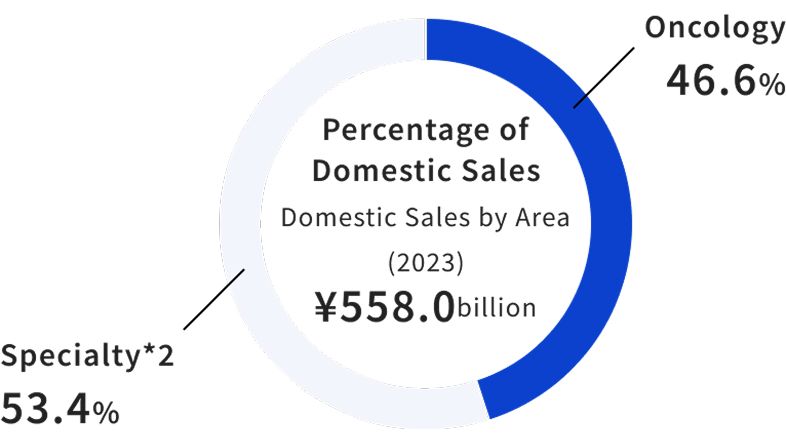
Domestic Product Sales by Category (2025)
In Japan, oncology products account for 52.2% of sales. Drugs in areas besides oncology, such as bone and joint diseases, autoimmune diseases, renal and neurological diseases, and hemophilia, are marketed in Japan as specialty areas.
- *2 Bone and joint, autoimmune diseases, renal and neurological diseases, and other areas
- *2 Bone and joint, autoimmune diseases, renal and neurological diseases, and other areas
As a research and development-oriented pharmaceutical company, we boast the No. 1 market share in Japan for antibody drugs. We are developing innovative new drugs for diseases for which effective treatments have not yet been established, addressing unmet medical needs.
67% of Chugai’s sales come from products covered by a system introduced by the Japanese government to accelerate the creation of innovative new drugs. The items covered by this system are new drugs that meet certain requirements and have not yet had generic versions released, showing that Chugai is developing, manufacturing, and marketing innovative new drugs.
-
Market share of antibody drug sales in Japan*1
No. 1
-
Proportion of sales from products that qualify for premium pricing:
67% (2024)
*1 Copyright © 2025 IQVIA. Calculated based on JPM 2024. Reprinted with permission. The scope of the market is defined by Chugai.

Q2Is Chugai an independent company?
In 2002, Chugai entered into a strategic alliance with Swiss company Roche.
Although Roche holds a majority stake of 59.89%, Chugai remains a listed company in Japan and continues to operate independently.
This strategic alliance has enabled the Roche Group to exclusively market their breakthrough medicines in Japan, particularly anticancer drugs, as well as strengthening the global development of Chugai’s own products.
About Roche
Established in 1896 and headquartered in Basel, Switzerland, Roche is the No. 1 pharmaceutical company in the world by revenue, with business operations in more than 150 countries. The Roche Group contributes to medicine through its two business segments of pharmaceuticals and diagnostics.
-
Advantage#1
Exclusive marketing of Roche products in Japan
-
Advandate#2
Global development of Chugai products
This unique business model has enabled Chugai to establish a stable revenue base and concentrate investment in more innovative technologies and drug discovery.
2025 Revenues
(compared to before the
alliance with Roche in 2002)
7.6times more
2025 Operating profit
(compared to before the
alliance with Roche in 2002)
23.3times more
For more information, click here

Q3What are Chugai’s strengths in drug discovery?
As a pioneer in the discovery of Japan's first developed antibody drug, we have an edge in biotechnology and are also focusing on research and development of mid-size molecule drugs as a new modality.
Since the 1980s, Chugai has been involved in the discovery of biopharmaceuticals. This experience has led to the creation of antibody drugs and the development of proprietary technologies. One type of biopharmaceutical, antibody drugs are expected to offer high therapeutic efficacy and reduced side effects by precisely targeting specific cells and molecules. In 2005, Chugai successfully launched the first antibody drug produced in Japan. Since then, we have delivered innovative drugs to the world, and our in-house products have received the U.S. Breakthrough Therapy *2 designation nine times.
Following antibody drugs and small molecule drugs, we are focusing on research and development of mid-size molecule drugs as the third drug discovery modality (classification of therapeutic approaches).
A drug discovery modality refers to a method of approach for creating drugs. Mid-size molecules are compounds that fall between small molecules and antibodies (macromolecules). They are characterized by their high specificity for binding to disease target molecules, and their abilities to bind to intracellular targets and be administered orally. Mid-size molecule drugs are expected to be a drug discovery modality that combines the advantages of small molecules and antibody drugs.
Breakthrough Therapy designations *2
9times
Six proprietary drugs developed through our domestic research and development have been designated as Breakthrough Therapies* by the U.S. Food and Drug Administration (FDA) a total of nine times. (Cumulative as of February 28, 2025)
Number of Countries with Global Approval for Chugai Proprietary Products
More than 110countries
Proprietary products researched and developed in Japan have been approved in more than 110 countries. (As of February 28, 2025)
Chugai Life Science Park Yokohama began operations as a hub for drug discovery research in 2023. We strive to accelerate Chugai's unique drug discovery engine through activities at both domestic and overseas research hubs and by promoting open innovation.

Q4What about Chugai’s use of Artificial Intelligence (AI) and other digital technologies?
We are promoting digital transformation (DX) and working to improve productivity and create value through the use of AI and other digital technologies.
In promoting company-wide DX, we have formulated the “CHUGAI DIGITAL VISION 2030.” We aim to continuously deliver innovative pharmaceutical products through the use of digital technology and to improve productivity across the entire value chain, from research and development to production and sales.
-
CHUGAI DIGITAL ACADEMY
A system developed to systematically train internal digital talent such as data scientists. We support the application of knowledge to real-world tasks through comprehensive lectures and on-the-job training (OJT).
-
Chugai Cloud Infrastructure (CCI)
CCI is Chugai’s company-wide digital IT platform. It provides enhanced security governance and efficient integrated operations management in a multi-cloud environment.
-
Smart Factory
We aim to achieve a smart factory that links and optimizes data between people and operations in the production of pharmaceuticals in factories.
-
AI Drug Discovery
We utilize AI to target molecules in drug development and to design molecules for antibody drugs and other drug discovery modalities. Our goal is to improve the success rate of drug development and the efficiency of the drug discovery process.
Selected as a DX Platinum Company
Chugai Pharmaceutical has been selected as a 2023-2025 DX Platinum Company for its outstanding and continuous DX initiatives, from among the DX brands selected by the Ministry of Economy, Trade, and Industry (METI) to accelerate DX in Japanese companies.

To learn more

Q5What kinds of people work for Chugai?
Chugai has a diverse workforce. We aim to foster an organizational culture that creates continuous innovation, and we have developed and are promoting a talent management policy to support this goal.
- *1 Consolidated, as of December 31, 2025
- *2 Managers with subordinates, based on Chugai Pharmaceutical Group enrollment in Japan, as of April 1, 2025
- *3 Based on the number of employees as of 2024 (individual company basis)
- *4 Based on non-consolidated financial reports, as of April 1, 2025
- *5 Cumulative across 2004 - 2024
To learn more

Q6What are Chugai’s social and environmental initiatives?
We will work together with various stakeholders to address healthcare challenges and protect the global environment.
Chugai aims to solve social issues through its business activities and develop together with society and local communities. Our sustainability initiatives, which create shared value with society, have garnered strong support from our stakeholders.
We engage in activities that consider CO2 emissions and procure electricity with a focus on preserving the global environment.
Sustainable Electricity Ratio
Domestic
Offices
100%
Energy sources
52.2%
reduction* in
CO2 emissions
*2024: compared to 2019
We aim to provide advanced patient-centric solutions.
We have established a system to provide solutions that carefully address the diverse needs of healthcare professionals in each area. We are committed to promoting multidisciplinary team care and regional healthcare coordination through various initiatives such as providing information, hosting study sessions, and conducting awareness activities in collaboration with government authorities. Additionally, we promote the proper use of pharmaceuticals and have established an industry-leading pharmacovigilance management system to ensure their safe use in clinical settings. We collect, evaluate, and provide drug safety information to support this effort.
Satisfaction of healthcare professionals
(Corporate Comprehensive Evaluation)
No.1*
*INTAGE Healthcare “Rep-i August 2024 Survey” (reprint prohibited); based on survey results for an overall company assessment targeting only physicians according to Chugai’s definition
Satisfaction of healthcare professionals
(Drug safety information provision)
No.1
in Japan*
* INTAGE Healthcare “2024 Drug Safety Information Needs Assessment Survey” (reprint prohibited); based on the total results of all respondents
The results of our activities have been evaluated by external evaluation organizations.




















