Utilization of real-world data (RWD)
By utilizing real-world data (RWD), we promote the efficiency and sophistication of the clinical development process of new drugs, the verification of drug values, and understanding of diseases, to provide an optimal treatment for each patient.
What is real-world data (RWD) and real-world evidence (RWE)?
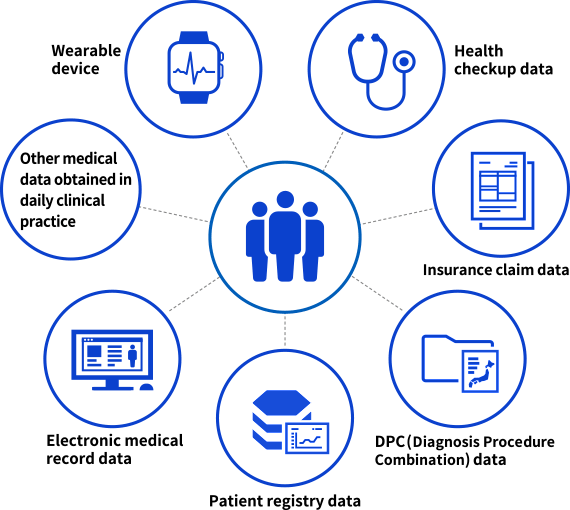
Real-world data (RWD) is a collective term for medical data gathered in daily clinical practice. RWD encompasses data collected from insurance claims, Diagnosis Procedure Combination (DPC), electronic medical records, health checkups, patient registries, and wearable devices. In recent years, advances in medical IT have enabled the acquisition and analysis of massive RWD.
Unlike data prospectively obtained with a predetermined purpose in clinical trials, RWD data are gathered from daily clinical practices and are very large in size, which provides an advantage of diverse patient backgrounds, albeit their inferior accuracy and reliability compared to those of clinical trials.
The scientific evidence obtained by analyzing real-world data is called real-world evidence (RWE).
RWD (conceptual diagram)
Expectations for utilization of real-world data (RWD)
Based on the analysis of medical data, including RWD, the following outcomes can be expected:
- Understanding diseases in basic medical research.
- Clinical development and application of drug approval.
- Early detection of health risks from pharmaceuticals and medical devices.
- Streamlining and optimization of diagnosis and treatment by physicians.
- Evaluation of and responses to government medical policies.
- Improvement in patient access to healthcare.
- Improvement of quality and efficiency of health management, lifestyle, and preventive services.
- Improvement in quality and efficiency of nursing services.
A proper understanding of clinical care and its results through the use and application of RWD is expected to improve the QOL of citizens and patients who provide data while securing a sustainable social security system.
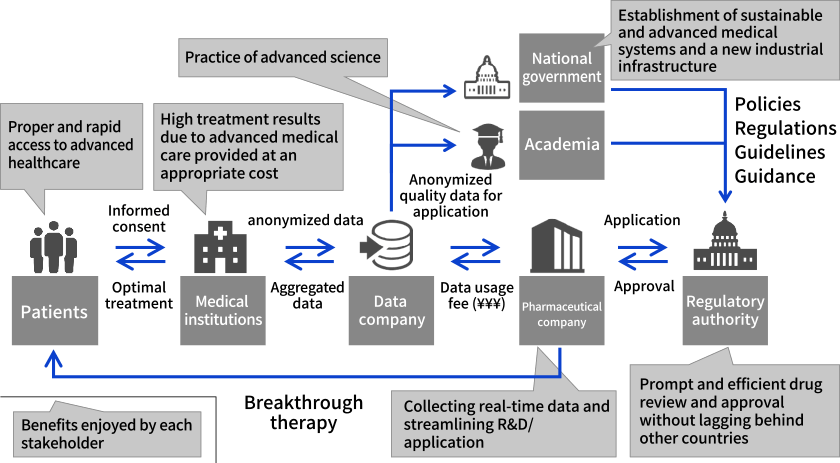
In Europe and the United States, RWD is increasingly used for approval applications for the clinical development of drugs, cost-effectiveness evaluations for insurance reimbursement, and drug price negotiations. In Japan, there is a debate over the need to establish an ecosystem that enables the collection and analysis of high-quality RWD that can be used for clinical development. To this end, relevant laws and guidelines are being prepared.
Ecosystem to utilize RWD (conceptual diagram)
Chugai’s policy on utilization of real-world data (RWD)
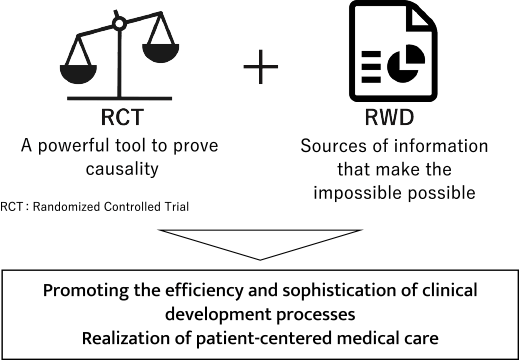
While clinical trials are conducted in the form of Randomized Controlled Trials (RCTs), Chugai aims to utilize RWD to increase the efficiency and sophistication of trial planning to assist in proving efficacy and safety in cases where it is difficult to conduct RCTs, such as rare diseases and diseases with a small number of patients.
In addition, we are working on the application of RWD for basic research to deepen the understanding of diseases and to verify drug values in daily clinical practice after approval.
Chugai’s policy on utilization of RWD
Chugai’s initiatives on real-world data (RWD)
The following are examples of Chugai’s initiatives in utilization of RWD:
Development of real-world data (RWD) analysis and research environment
Chugai develops an environment in which medical data, including RWD, are used in a timely and appropriate manner in compliance with the privacy policy.
[Features of Chugai’s environment to analyze RWD]
- Analysis platform with enough performance to process big data is available.
- A transparent operation system has been established by adopting data security requirements and rigorous rules for privacy protection.
- Guidelines for using RWE obtained from RWD for drug approval applications have been developed and appropriately implemented.
Cross-departmental RWD collaboration system
The cross-departmental collaboration system has been established, and the entire company is working to promote the efficiency and sophistication of RWD analysis technology, and to utilize RWD to establish the efficacy and safety of drugs.
Joint research with National Cancer Center Japan
We have conducted an exploratory study of efficacy and safety using anonymized electronic medical record information with National Cancer Center Japan.
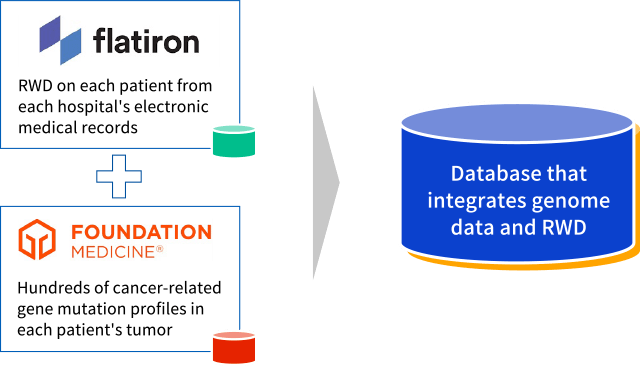
Collaboration with Roche: Flatiron and Foundation Medicine
With a strategic alliance with Roche, Chugai has the advantage of easy access to RWD, and the platforms owned by Roche. In collaboration with Roche, the world leader in personalized medicine, we aim to maximize the use of RWD. This will allow us to integrate clinical data from Flatiron’s electronic medical records as well as the genetic mutation data profile of cancer patients from Foundation Medicine.
Example of cooperation with Roche: Integration and utilization of RWD and genomic data
CAREERS
Chugai is looking for talent to help us harness digital technology to become a leading innovator in the healthcare industry.