Drug Safety Core Themes and Initiatives
Key Focus Areas
Chugai is advancing drug safety by prioritizing the following three key strategic areas.
1. Elucidating adverse reaction mechanisms using non-clinical and clinical data, and translating findings into safety measures
In collaboration with partners such as Roche and academic institutions, we analyze patient-stratified safety profiles using real-world data and biomarkers. We also investigate the relationships between safety outcomes and patient background factors, including genetic characteristics and comorbidities, to generate evidence that supports optimal treatment selection tailored to individual patients.
2. Generating safety data early in the post-marketing phase to enhance predictability and prevent serious adverse events
From the early stages of development, we assess and predict potential safety risks and establish comprehensive risk management plans. By identifying gaps in available data, we contribute to improving the predictability of adverse reactions and preventing their progression to serious conditions. By generating safety data early in the post-marketing phase, we aim to ensure that patients can receive treatment more safely.
3. Promoting appropriate use to deliver safety and reassurance to patients and healthcare professionals
We rapidly and comprehensively collect and analyze safety information on our products from around the world, and through continuous monitoring, maintain up-to-date product safety profiles. By combining this information with digital technologies and the needs of clinical practice and patients, and providing it as optimal safety measures, we promote the appropriate use of our products in real-world clinical settings.
Specific Initiatives
Below, we introduce specific initiatives based on the three key focus areas.
1. Collection and management of safety information and risk assessment
We collect safety information observed in real-world clinical practice that cannot be obtained through clinical trials, via reports from medical institutions as well as information from scientific literature and academic conferences. In addition, we proactively collect safety information through post-marketing surveillance activities, including all-case surveillance and database studies.
Safety information collected from medical institutions is assessed using multifaceted approaches, including epidemiology. By continuously reviewing safety profiles, we promote post-marketing pharmacovigilance activities. The results obtained are shared with medical institutions and published through academic conferences and scientific journals.
Furthermore, for many anticancer agents and innovative biopharmaceuticals, we implement broader and more stringent management measures – such as distribution control and confirmation of conditions of use – and utilize these measures in safety evaluation and risk mitigation.
2. Transition from reactive to predictive and preventive safety measures
In addition to conventional reactive post-marketing pharmacovigilance activities, in which risks are assessed and safety profiles are developed on the basis of safety information collected from medical institutions, we are strengthening efforts to shift toward more predictive and preventive safety measures. By formulating and validating hypotheses based on diverse insights from the early stages of clinical development, we aim to predict individual patient risks after approval and to prevent the occurrence and progression of adverse reactions.
Through these preventive post-marketing pharmacovigilance activities, we strive to ensure that patients can focus on their treatment with confidence from the early post-launch phase.
3. Safety communication
In collaboration with patient advocacy groups, we provide information that patients want to know and information that we want patients to know in real-world treatment, through a variety of solutions including educational materials.
To enable the rapid provision of information tailored to patient characteristics, we have been operating survey and safety database tool since 2016. Following a system renewal, healthcare professionals can now access adverse event information related to our products at any time via our dedicated website for healthcare professionals. We have also improved the user interface to ensure intuitive usability. Through continuous enhancement of these information delivery platforms, we strive to provide safety information more smoothly and effectively to each individual patient.
Cycle of Safety Information Collection, Accumulation, and Provision at Chugai
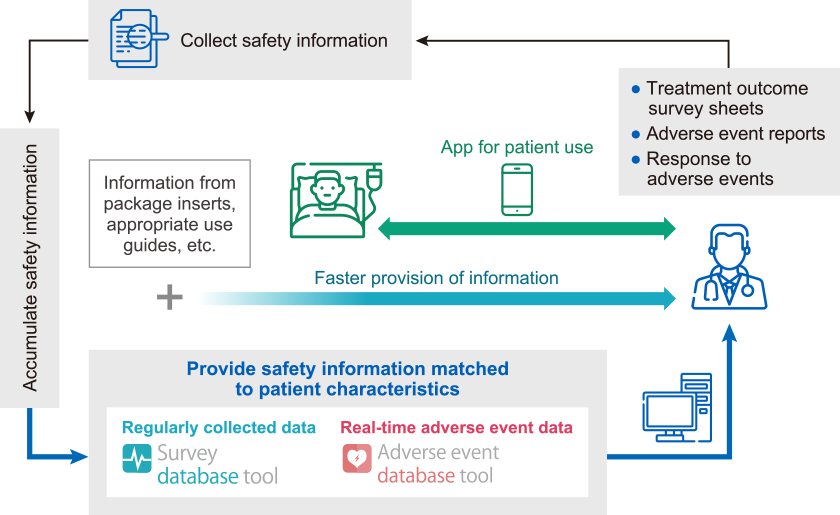
At Chugai, we continuously strengthen initiatives to facilitate smooth communication between patients and healthcare professionals and to enable patients to receive treatment with greater confidence and peace of mind.