R&D Process
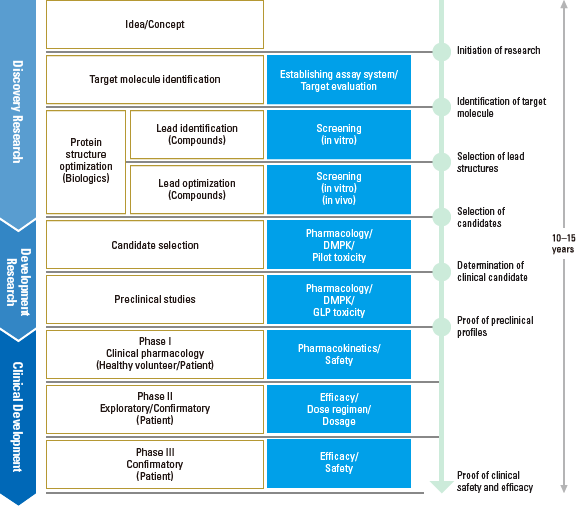
Process and Milestones of Drug Development
It takes an average of more than 10 years to develop a single new drug. The following research and development process will lead to the emergence of a new drug.
Glossary
Business Process
Please refer to the following page for information on the business process that Chugai uses to create innovative medicines.