Production Technology
Our pharmaceutical technology and production functions play a range of roles, from establishing manufacturing processes for investigational candidates – whether developed in-house, in-licensed from Roche, or originating elsewhere – to ensuring the stable supply of these products. By ensuring continuous evolution of our technologies and working to maintain and strengthen the supply chain, we aim to become a top innovator maintaining the trust of patients and healthcare professionals.
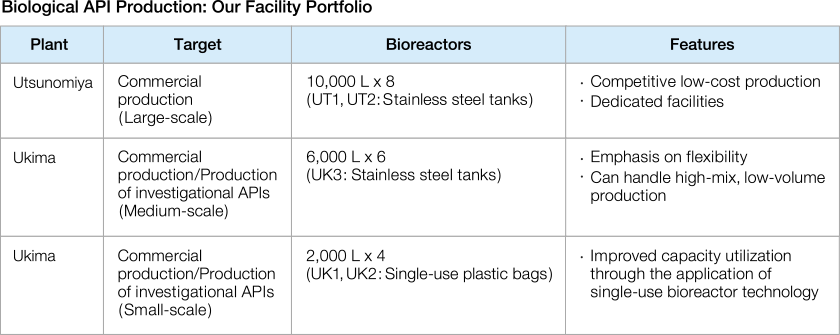
Improving Flexibility and Speed
In its pharmaceutical technology and production operations, Chugai is aiming for simultaneous development of multiple products for the quickest launches possible. Specifically, the Ukima Plant has achieved a significant increase in capacity utilization by employing single-use plastic bioreactors. For development candidates based on next-generation antibody engineering technologies, the plant has also begun full-scale operation of UK3, an antibody API facility capable of high-mix, low-volume production from late-stage development to initial commercial products. At the Utsunomiya Plant, we have increased production flexibility by installing tray fillers that can handle filling of liquid medicines without making line changes or modifications, regardless of the syringe type.
Evolution of Pharmaceutical Technology and Production Functions through DX
Chugai has implemented initiatives to help realize smart factories that can accelerate in-house drug discovery and respond flexibly to changes in the environment. Under the concept of “digitally transforming production operations to increase productivity and add high value to human capital,” we are partnering with IBM Japan, Ltd. to link and optimize people and operational data.
Through DX focused on human functions, we are driving improved productivity and reliability and innovation in workstyles. Implementation will begin first at the Ukima Plant’s UK3 facility, which will serve as a model, with rollout to other production bases planned for the future.
Evolving Supply Chain Management
To minimize risks related to the supply and price of raw materials, we promote the globalization and establishment of two supplier bases for raw materials and intermediate products in tandem with the globalization and establishment of two production bases for each finished product.
Thorough Quality Assurance
Quality assurance functions have diversified in recent years in response to the increasing complexity of supplying products and accelerated development with the introduction of the fast-track review system to support the early launch of innovative new drugs. In view of these trends, Chugai is working to further strengthen GMP* management oversight to promote more rigorous and high-level quality assurance. As part of these efforts, Chugai promotes the building and operation of a world-class system for pharmaceutical quality management.
- * Good Manufacturing Practice: Standards for pharmaceutical production management and quality control
