Innovating Pharmaceutical Packaging to Protect Patients and the Planet: Chugai's Challenge to Implement Recycled Plastics and Biomass-Based Materials
- R&D
- Sustainability
- Sustainable Healthcare
- Global Environment

Pharmaceutical packaging is far more than a simple container. It serves a vital mission: safeguarding drug quality, supporting improved usability for patients, and reducing the burden on the global environment.
Since 2019, Chugai Pharmaceutical has focused on four packaging formats that account for particularly high volumes of plastic use — press-through pack (PTP) sheets, plastic bottles, aluminum pouches, and syringe blister trays — accelerating the transition to eco-friendly materials. Most notably, the implementation of biomass plastics in PTP sheets for large capsules (size 0) that are difficult to form has been recognized as a pioneering achievement in the industry, earning both the Japan Packaging Contest 2025 award and the WorldStar Global Packaging Awards 2026.
We sat down with Kenichi Sakai and Hiroaki Oie of the Formulation Development Department, who have led this project, to learn about the technical breakthroughs behind the work and the culture of challenge that defines Chugai.
Packaging Actually Shapes Drug Quality and the Patient Experience
— What role does packaging play in pharmaceuticals?
Oie Pharmaceutical packaging serves three core functions: ensuring quality, improving usability, and providing information. Drugs cannot be distributed or stored in their unpackaged state — it is only by protecting them from external factors such as light and moisture that quality can be maintained. Labeling that correctly identifies each medication is also an essential function of packaging.
Sakai Our department is specifically responsible for designing for quality assurance and usability. How easily and safely can a patient remove a medication from its packaging? We see this as a critical part of the "patient experience" — one that directly affects treatment adherence. That is why we actively seek input from patients and healthcare professionals, including through direct dialogue with patient advocacy groups.
— Is it possible to balance environmental responsibility with quality and usability?
Oie People often raise the concern that environmental considerations might compromise quality, but we believe that with the right design, both can be achieved. At Chugai, the fundamental premise is that any new eco-friendly material must deliver quality equivalent to that of conventional packaging. From there, we work to leverage the unique properties of these new materials in our designs.
Why Eco-Friendly Pharmaceutical Packaging Matters Now
— Plastic regulations are tightening worldwide. What is happening in the pharmaceutical industry?
Oie Japan's Plastic Resource Circulation Strategy, formulated by the government in 2019, sets targets for 2030 that include expanding plastic recycling and scaling up the adoption of biomass plastics to a maximum of approximately two million tons. In Europe, regulatory pressure on packaging is intensifying, led by the enactment of the Packaging and Packaging Waste Regulation (PPWR). The hurdles are exceptionally high — encompassing not only safety and quality assurance but also a complex array of regulations and legal requirements. Yet the urgency is undeniable: extreme weather events driven by global warming and the impact of ocean plastics on ecosystems are pressing challenges on a global scale. Against this backdrop, with growing demands to reduce both CO₂ emissions and plastic usage, Chugai is tackling these issues head-on with the ambition of becoming a role model for the world.
Chugai's Eco-Friendly Packaging Roadmap and a Culture of Challenge
— Can you walk us through the milestones of this initiative?
Oie Starting in 2019, we zeroed in on the four packaging formats with the highest plastic usage and took a focused approach:
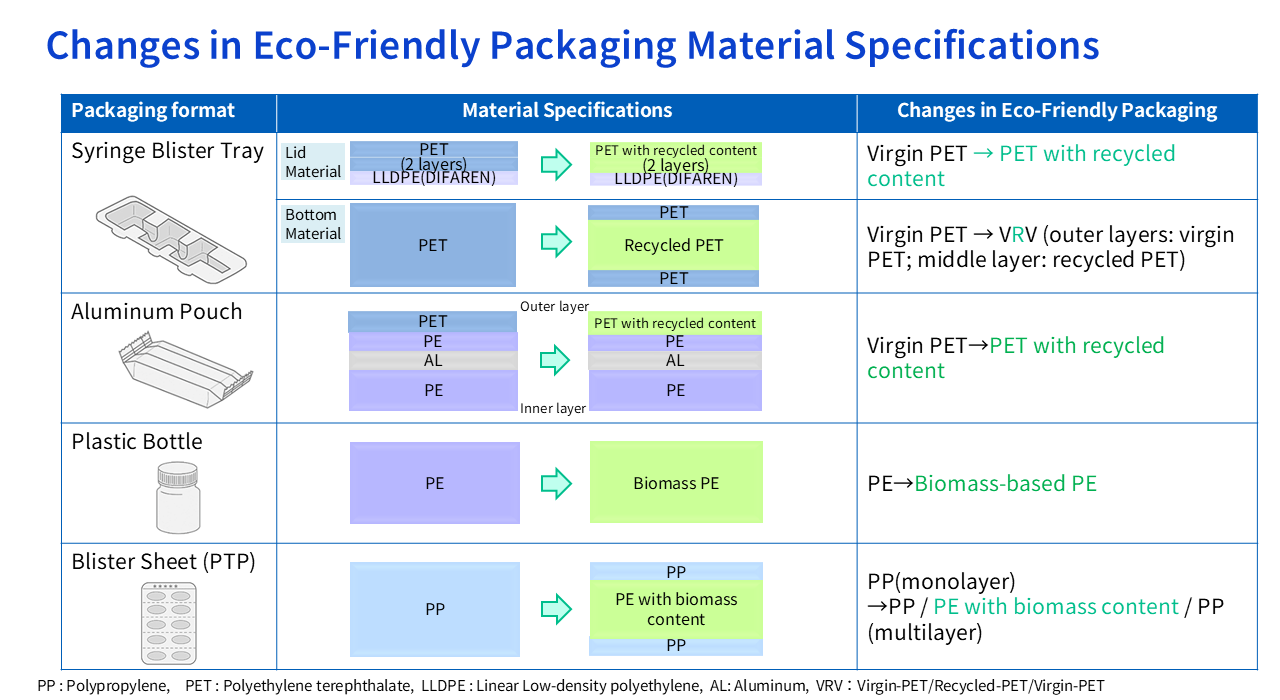
・2022: Introduced recycled plastics (recycled PET) into aluminum pouches and syringe blister trays.
・2023: Began implementing biomass plastics (biomass-based PE) in PTP sheets and plastic bottles.
・2030: We have set a goal to assess the feasibility of eco-friendly packaging materials across all products and pursue phased implementation.
— What was behind this industry-leading decision?
Oie What made it possible was that a bottom-up proposal from the front lines — team members saying, "We want to explore eco-friendly packaging materials" — aligned perfectly with a top management message of "addressing environmental issues in earnest," and the two converged at exactly the same time. Back in 2019, there were very few examples of eco-friendly packaging implementation anywhere in the industry, but there was a strong conviction within the company that "this is something Chugai should do."
Sakai Once the project launched, the team on the ground was highly motivated. As researchers, we found tremendous purpose in the opportunity to simultaneously advance Chugai's mission of contributing to patients and caring for the global environment. With strong backing from leadership, team members proactively shared ideas and worked together toward a common goal — that distinctly Chugai culture is what drove the speed of this implementation.
Science-Based Decisions: Constraints and the Logic of Material Selection
— In this initiative, you use recycled plastics (recycled PET) and biomass plastics (biomass-based PE) for different packaging components. What was the scientific rationale?
Oie As a starting point, there are various approaches to reducing environmental impact, including reducing the volume of materials used. From a material-selection perspective, there are two main strategies: recycled plastics, which reuse post-consumer plastic, and biomass plastics, which are derived from renewable, plant-based raw materials.
— What constraints did you face?
Oie Among recycled plastics, mechanically recycled materials — the most widely used type today — carry inherent risks of impurity contamination and quality variability that cannot be entirely eliminated. Because pharmaceuticals demand the highest levels of safety and quality, we made the decision to limit recycled PET to secondary packaging — aluminum pouches and syringe blister trays — where the material does not come into direct contact with the drug.
For primary packaging — PTP sheets and plastic bottles, which are in direct contact with the drug — we needed to select a material that allows more rigorous quality control. That led us to adopt biomass-based PE, which is derived from plant-based raw materials. Although its origin is plant-based, the molecular structure of biomass-based PE is identical to that of conventional petroleum-derived PE. Through appropriate selection and evaluation, we determined that it could meet the quality standards required for pharmaceutical packaging.
Case Study: The Technical Difficulty of Eco-Friendly PTP Forming for Large-Format Drugs
— Tell us about the project that posed the greatest technical challenge.
Sakai It was the implementation of biomass-based PTP for an anticancer drug, which we began exploring in 2020. This anticancer drug is an oral capsule formulation. Previously, it had been packaged using conventional bottle packaging in which the drug was filled into large bottles. Based on usability needs from both healthcare professionals and patients, we decided to switch from conventional bottle packaging to PTP packaging, which is better suited to dosage quantities. We needed to develop a sustainable, user-friendly packaging solution that would meet the needs of both healthcare professionals and patients while fulfilling our environmental responsibility.
Oie This drug uses one of the largest capsule sizes in our portfolio — large capsules (size 0) — which made the packaging extremely difficult to form.
— Why is the combination of size 0 and eco-friendly materials so challenging?
Sakai PTP sheets are formed by applying heat to a film to shape it into "pockets." To accommodate large capsules (size 0), the film must be stretched very deeply and over a large area.
The multi-layer film containing biomass-based PE that we adopted is physically "softer" than conventional single-layer PP film. This softness became a liability: during forming, the film would overstretch, causing the top and side walls of the pocket to become extremely thin — a phenomenon known as "insufficient film thickness" (see diagram below). This was a critical problem, as it compromised the protective function essential to safeguarding drug quality.
— How did you overcome this physical challenge?
Oie To get into the technical details, we used a method called plug-assist pressure forming. Whereas forming conditions are typically adjusted using around three main parameters, we expanded the adjustment to five parameters. Through repeated prototyping and evaluation on production equipment, we identified optimal forming conditions that equalize film thickness even for large capsules (size 0).
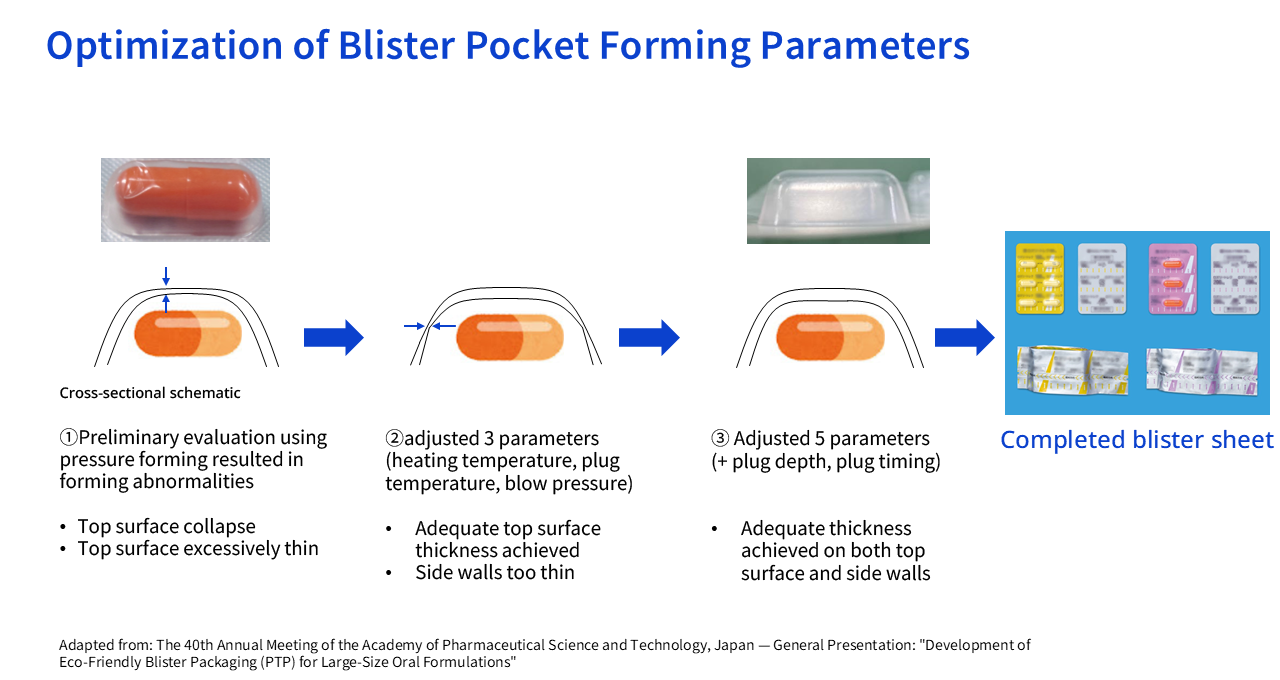
Sakai On top of that, there was another obstacle: "pocket collapse." Due to the combination of material properties, pocket geometry, and transport conditions, the load from transport rollers caused the formed pockets to collapse. We addressed this by redesigning the roller groove profile to match the characteristics of the material, enabling stable transport. The development of this anticancer drug packaging led to an international technology award at the WorldStar Global Packaging Awards 2026. Our packaging was recognized globally as a sustainable packaging solution that simultaneously achieves reduced environmental impact through the use of biomass plastics and recycled plastics, stable forming quality for a difficult-to-form size, and improved usability for patients and healthcare professionals.

The Next Challenge
— What does the future hold?
Oie We continue to advance research and development on eco-friendly packaging materials with even higher functionality. This direction will not change. We will keep building on our research and implementation efforts to deliver products with greater convenience for patients and contribute to a better society.
— Any final message for our readers?
Sakai Chugai is deeply committed to packaging technology R&D, and we place great value on creating packaging that "no one has done before" and delivering it to patients and healthcare professionals. There are very few opportunities to study packaging technology as a specialty at university, but it is a truly enjoyable and rewarding field of research. I would love for more people to know that.
Oie I see packaging as a vital interface connecting drugs, patients, and society. The steady accumulation of day-to-day technology development leads to higher quality healthcare and greater environmental responsibility. With that conviction, I will continue working to deliver value to patients, healthcare institutions, and society through the development of eco-friendly packaging materials.
Kenichi Sakai (Formulation Development Department)
Sakai joined Chugai Pharmaceutical in 2000 and began his career in oral formulation development. He built extensive experience as a professional leading technology development and currently heads the packaging research and development organization. With patient convenience as his starting point, he leads an integrated strategy encompassing advanced packaging material functionality, sophisticated packaging design, reduced environmental impact, and cost optimization — all aimed at maximizing product value and driving sustainable corporate growth.

Hiroaki Oie (Formulation Development Department)
Oie joined Chugai Pharmaceutical in 2008. He has been responsible for packaging design, technical evaluation of development compounds in oral and injectable formulation development, as well as packaging improvements for existing products and technology transfer to manufacturing sites. He currently focuses on advancing technology development in packaging and regulatory submission support. Throughout his career, he has driven the adoption of eco-friendly packaging materials — including biomass-based and recycled materials — in commercial product packaging.



