Feature: Delivering Safe, Reliable Drugs: Chugai's Uncompromising Pursuit of Drug Safety
Leading the Industry in Safety Evaluation of Biopharmaceuticals and New Anticancer Agents
All-case registration surveillance is often conducted after obtaining marketing approval for innovative new drugs such as anticancer agents or biopharmaceuticals. In this surveillance, medical institutions and all patients using the drug are registered, and wholesalers and dispensing pharmacies are given explanations before the product is used. Moreover, thorough, wide-scale monitoring of patient use and drug safety is conducted, including verifying the expertise of medical institutions and pharmacists and their ability to deal with adverse drug reactions when they occur. Ahead of other companies, Chugai conducted large-scale all-case registration surveillance for the biologic treatments Avastin®, Tarceva® and Actemra® as well as anticancer agents with novel modes of action. We have presented the results of this surveillance and the analysis of the extensive data collected at scientific conferences and published our findings in academic papers. Consequently, Chugai is now recognized as an industry leader in this area.
Even when all-case registration surveillance is not required, we carry out equivalent safety monitoring plans when we deem it necessary. These efforts consume considerable resources, but we have a mission to ensure the safe and appropriate treatment of patients by securing the safety of our pharmaceutical products.
Chugai collects safety information on approximately 100,000 cases each year, among the most for Japanese pharmaceutical companies, and conducts high-quality evaluations from a medical standpoint. We record the results of these evaluations in a global database and promptly disclose them to regulatory authorities in Japan, Europe, North America and Asia as well as to medical institutions. Aside from having a large volume of safety information, our Drug Safety Division is staffed with licensed medical practitioners with extensive clinical experience who conduct expert medical evaluations.
Moreover, strengthening our ability to analyze safety data from an epidemiological standpoint is essential for ensuring drug safety. At Chugai, we have been working to improve the precision of analysis through a specialized internal group in charge of epidemiology functions, and we actively cooperate with specialist companies and others to help upgrade Japan’s epidemiological database.
Preparing and Communicating Pharmaceutical Risk Management Plans: Our Commitment to Patients and Medical Institutions
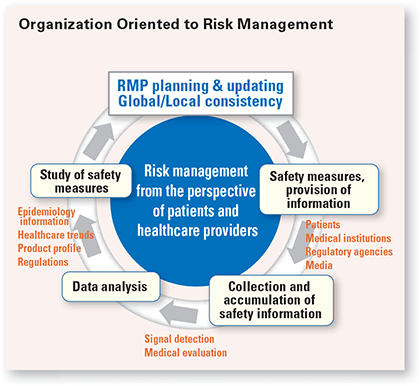
Pharmacovigilance refers to the science and activities related to identifying, assessing, understanding and preventing adverse drug reactions or other problems related to the drug. Awareness of the importance of pharmacovigilance has risen rapidly in recent years, and programs around the world are more active than ever. As a result, pharmaceutical rules and regulations are also becoming more stringent. Companies are required to collect a wider range of safety information, including information obtained in patient support activities, establish globally standardized safety management systems, and increase transparency by promoting direct communication with medical institutions and patients.
Given this situation, Chugai has further enhanced its world-class safety management system to establish a plan-do-check cycle in pharmacovigilance activities, and to conform to the new pharmaceutical supervision systems of various countries. As one outcome of that, we began drawing up pharmaceutical risk management plans (RMP) for five of our products in 2012, ahead of our competitors, and have already started applying them. Drafting and implementing RMPs became legally mandatory in Japan in April 2013. However, we proactively began applying these measures because of our firm commitment to help patients and medical institutions use our products more appropriately. We fulfill this commitment by gaining an understanding of RMPs, in which we have formulated methods for monitoring adverse drug reactions and ways to minimize risk with the recognition that pharmaceuticals constitute a balance between therapeutic benefits and side effects. Chugai will continue to carry out various measures based on its commitment, including establishing a signal detection system, conducting evaluations with a high level of expertise and making speedy decisions on measures to ensure safety.
Providing Safety Information in Line with Global Standards
Chugai is taking advantage of its membership in the Roche Group to standardize the evaluation and dissemination of safety information worldwide, and to conform to new global safety standards. Recently, we have established innovative pharmacovigilance-related interactive communication protocols with other partner companies, and are making arrangements for their smooth operation. In addition to standardizing safety evaluations for each product and sharing information on adverse drug reactions, we have established a worldwide framework for speedy decision-making on safety measures and methods of response in coordination with Roche.
Moreover, we compile information on, and typical examples of, potential risk factors for the inherent adverse drug reactions of each product. However, simply possessing information is meaningless. Only when that information is actively conveyed and publicized does it have meaning for patients and medical institutions. That is why we distribute patient-oriented leaflets explaining adverse drug reactions and post the information on the Chugai website, while our medical representatives respond to inquiries from medical institutions individually. These activities help to reduce the incidence and aggravation of adverse drug reactions by creating an environment for treatment that takes high-risk patients into consideration.
We formulate and implement these initiatives not just to conform to the new regulatory systems but because we want to improve the treatment of patients by ensuring that our products are used appropriately. We will continue to provide truly valuable data quickly and accurately as part of our contribution to healthcare worldwide.

In the pharmaceutical industry, safety initiatives are in the spotlight like never before. With strict demands for safety transcending national boundaries, measures to secure patient safety must extend from the early stages of product development to the post-marketing stage. Moreover, I strongly feel that better communication with patients and medical institutions is essential for gaining trust and maintaining a high level of transparency.
Pharmaceuticals constitute a balance between therapeutic benefits and side effects. For that reason, accurately assessing and keeping the risk of adverse drug reactions within a fixed range and taking proactive risk management measures are critically important in patient drug therapy. This approach allows us to offer patients the most value from our medicines, and is in fact a fundamental part of our mission. At Chugai, we have turned the various events and experiences that have occurred with pharmaceuticals up to now into a source of strength, and are focusing our full efforts on drug safety initiatives to fulfill our accountability to patients and medical institutions.
Common to all of these initiatives is that we think about what standards are truly necessary to ensure drug safety, not just to meet regulatory standards, and follow them without compromise. We will not limit ourselves to in-house activities, but expand the scope of our efforts to include the industry and medical institutions. We also want to communicate appropriate, accurate and timely drug information that is truly meaningful in patient treatment. I believe that is Chugai’s mission as an industry leader.
Yoshiaki Ohashi
General Manager of Drug Safety Division

