The first 10 years since Chugai began the strategic alliance with Roche in 2001 were the so-called first stage. During that period, to help eliminate the drug lag we focused on bringing numerous in-licensed Roche products already on the market overseas to patients in Japan as quickly as possible.
Meanwhile, we steadily built global development infrastructure for providing our own products to patients around the world.
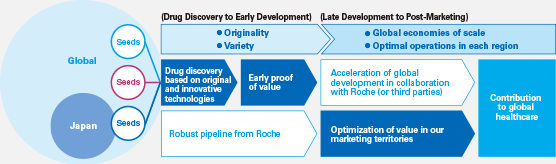
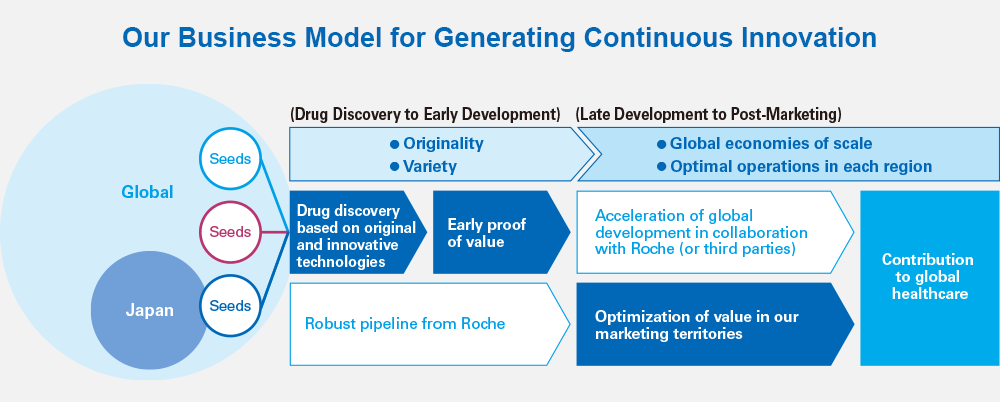
We decided to conduct Chugai-managed global development of SA237 and nemolizumab in conjunction with the previous mid-term business plan, ACCEL 15. It was tough going for both development projects, on which we focused our efforts for approximately three years, but we successfully completed global clinical studies and out-licensed the compounds, which will lead to maximization of product value.
We learned a lot through the first Chugai-managed global development projects spanning Asia, the United States and Europe. Recently, in an effort to quickly obtain early PoC*1 for products from Chugai research, we appointed a Chief Medical Officer (CMO),*2 established the Translational Clinical Research (TCR) Division,*3 and strengthened overseas operating bases by hiring local talent, among other measures, to evolve our development system globally.
Under IBI 18, Chugai is focused on improving the process for proving medical and economic value. We concentrate resources on the phase leading up to PoC. In addition to PoC data, if we can generate added value that leads to proof of medical and economic value, our licensees can assess the value of our medicines more precisely and smoothly execute more effective phase III clinical trials.
Based on that idea, we have launched a specialized unit that is currently advancing initiatives for proving value in the early stages of development. Even in our out-licensing strategy, we want to contribute to patients around the world by maximizing the value of our medicines through innovative approaches.