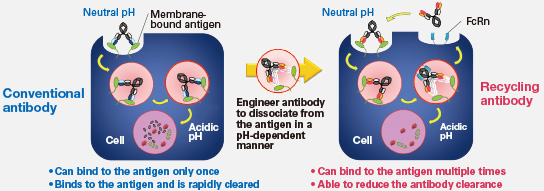
SA237 is an anti-IL-6 receptor antibody, and was created using Chugai's innovative proprietary recycling antibody technology. Unlike ordinary antibodies, which bind to a target antigen once and then are cleared from the body, the recycling antibody is designed to bind to the antigen repeatedly. Since the serum level of recycling antibodies is maintained for a longer time than that of ordinal antibodies, effects can be expected for the recycling antibody with a longer dosing interval and smaller doses.
With this technology, Chugai created the anti-IL-6 receptor antibody, SA237. A sustained effect with subcutaneous injection of small doses was confirmed, in line with the assumptions in basic research. Based on that finding, Chugai started full-scale clinical development of SA237.
Lifecycle Leader (currently Lifecycle Strategic Leader) Dr. Higashi recalls, “When we thought about the value that SA237 can bring to patients and its potential as a future growth driver, we wanted to conduct its global development. We had an obligation to bring this revolutionary new drug to the world.” Thus began a new challenge for SA237.
In 2011, a paper presented by Dr. Takashi Yamamura of the National Center of Neurology and Psychiatry influenced SA237’s destiny.
Neuromyelitis optica (NMO) is designated as a rare and incurable disease for which no viable treatment options have been established. Dr. Yamamura demonstrated that since an antibody against a protein called aquaporin-4 (AQP4) is expressed at high levels in NMO patients, blocking the function of IL-6, which is essential for production of the AQP4 antibody, could improve the symptoms.
Clinical studies of administration of an anti-IL-6 receptor antibody also started, and in 2013 data showing clinical efficacy was reported for the first time. In the same year, two research groups in Germany reported the effectiveness of inhibition of IL-6 signal transduction in NMO patients.
Dr. Higashi and her team deepened their contact with Dr. Yamamura, who had an existing cooperative relationship with Chugai in the neurology field, and explored the possibility of applying the higher added value of SA237 to the treatment of NMO, an area of unmet medical need. This drastically changed the direction of global development to bringing SA237 to patients worldwide.
At the time, Chugai did not have sufficient experience for the trilateral development among Japan, the United States and Europe. The team began studying the idea of boldly conducting phase III clinical trials globally, and immediately went to work preparing a clinical trial protocol and creating the structure for implementation.
The team also prepared meticulously for consultation with regulatory authorities in close cooperation with subsidiaries Chugai Pharma USA and Chugai Pharma Europe. However, different trial methods were specified by U.S. and European regulatory authorities, and after much agonizing, it was decided to conduct two trials to meet the requirements of both. To promote patient enrollment, the countries covered by the clinical trials were expanded beyond the United States and Europe to include more than 20 countries overall.
In June 2016, Chugai and Roche concluded a license agreement for SA237 with a focus on the CNS field. With this overseas out-license, Chugai gained positive recognition for its global development and created a path for delivering SA237 to patients around the world.
The SA237 project team accumulated valuable experience in overcoming a series of challenges and struggles. The project not only deepened knowledge in conducting Chugai-managed multinational studies, but also presented a good opportunity to directly understand global development by conducting clinical trials in various countries beyond the United States and Europe.
The knowledge we have gained is already spurring change in various business schemes in the Company, and is a driving force accelerating the enhancement of Chugai's efforts in translational clinical research. Moreover, we reconfirmed the importance of explaining business value objectively at the early PoC stage to facilitate out-licensing overseas.
Therefore, in IBI 18 we are working from the discovery stage to strengthen the integrated development and commercialization plan (IDCP)*4 process for proving medical and economic value.
“IL-6 is a very important cytokine*5 related to a variety of pathologies. We will continue to consider adding new indications for SA237,” says Dr. Higashi. The team is continuing global development and is already looking toward the next stage.
In retrospect, Dr. Higashi and Mr. Yamada (currently Lifecycle Leader) believe that they were able to overcome the project’s numerous issues because they constructively faced the need to overcome those issues regardless of difficulty, supported by their commitment to bringing SA237 to patients around the world.
NMO
NMO is a neurological autoimmune disorder characterized by severe optic neuritis and transverse myelitis. It occurs in about 0.3 to 4.4 out of every 100,000 people, and many patients experience repeated relapses and develop visual loss and gait impairment, along with chronic pain. In Japan, NMO had been equated with multiple sclerosis (MS), but in 2005 the overexpression of the anti-AQP4 antibody was confirmed, and an anti-IL-6 receptor antibody was subsequently found to be effective.
Recycling Antibody and SA237
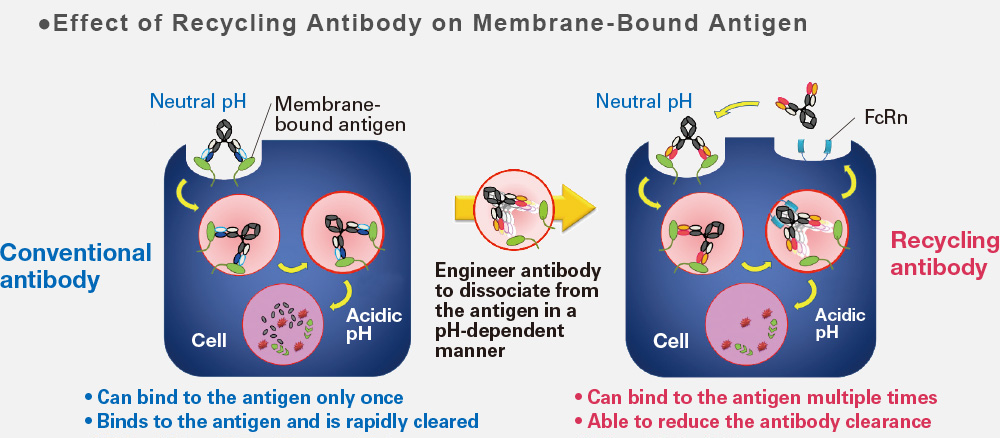
Chugai's proprietary recycling antibody technology, applied in SA237 is designed to dissociate from the antigen under acidic conditions, and can bind to the antigen multiple times. As a result, it shows significantly longer plasma persistence and effective duration. SA237 is receiving increasing attention worldwide as a potential treatment for NMO, which does not yet have any approved remedies.
■ Effect of Recycling Antibody on Membrane-Bound Antigen