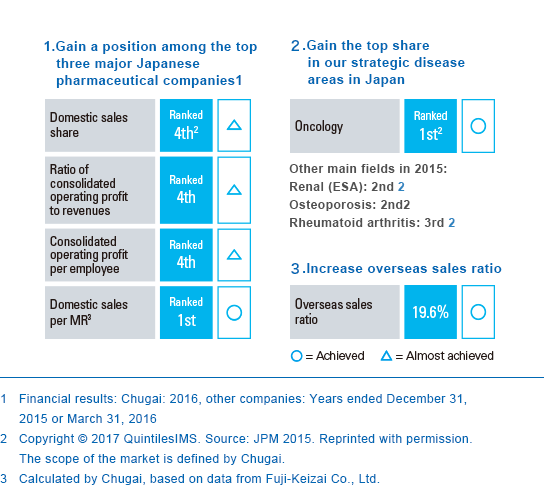
To achieve the objectives of IBI 18, to realize our goal of becoming a top pharmaceutical company and to grow globally in the future, we must accelerate selection and concentration of our resources and further enhance our global competitiveness. We are making solid progress toward our quantitative targets for becoming a top pharmaceutical company, but as I mentioned earlier, changes to the drug pricing system and other factors are making the industry environment and competitive landscape increasingly unclear.
In these circumstances, Chugai has clearly positioned the Japanese market as a source of stable revenues and the market outside Japan as a source of growth through the global expansion of products from Chugai research. By enhancing each of these pillars, we will build a strong foundation for future growth.
Our priority agenda objectives for 2017 are “focus on global and domestic growth drivers,” “creation of engineered antibody projects and development of middle-molecule technologies,” and “execution of system reform to provide solutions.”
For our growth drivers, we will continue to concentrate resources on emicizumab and atezolizumab (RG7446). We plan to file applications in and outside Japan for regulatory approval of emicizumab as a potential treatment for hemophilia A. Aiming for the speediest possible launch, we have started to set up of the production system for emicizumab and have begun construction of a biologic API production facility at the Ukima Plant to meet expanding global demand. Atezolizumab is a cancer immunotherapy, a category in which intensifying global competition is expected, and we will concentrate expenditures and personnel on this project to compete successfully in terms of speed.
In discovery research, our goal is to move two engineered antibody projects based on our proprietary technologies into clinical development during IBI 18. In middle-molecule projects, as the creation of a candidate compound moves closer to becoming a reality, we will evolve our compound library and resolve technical issues with the aim of creating an early preclinical project. In basic research, a new laboratory will be established and begin operation from April 2017 at IFReC, our comprehensive collaboration partner.
As for system reform to provide solutions, it is important to establish a system for the global market that is efficient and adapted to conditions in each country. We will quickly implement reforms led by overseas subsidiaries. In Japan, healthcare system reforms will place the healthcare delivery system under the leadership of prefectural governments from 2018. Ahead of these reforms, we will make organizational changes in April 2017 in marketing and sales, medical affairs and drug safety to offer greater expertise while responding precisely to the different circumstances in each region.
(See “About the New System to Provide Solutions in Japan” on page 35 for details about these new organizational reforms.)
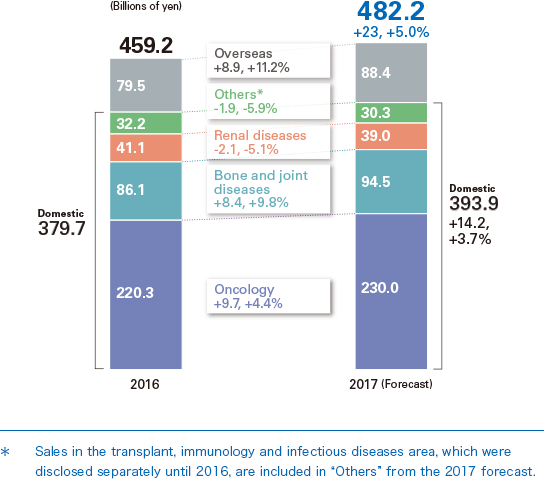
In 2017, we are anticipating revenues of ¥520.5 billion and Core operating profit of ¥92.0 billion, both record highs, driven by the solid growth of our major products in Japan and worldwide, including new products such as Alecensa.
Numerous regulatory filings are planned for 2017. Specifically, these will include filings for approval of emicizumab (for patients with inhibitors) in and outside Japan, for Alecensa in first-line treatment outside Japan, and for two expected indications for atezolizumab in Japan. Many more applications are planned for 2018 and later.
Our goal in IBI 18 is to transform into a globally successful company. The many products with regulatory filings scheduled during IBI 18 will begin contributing to sales after the plan is completed, but we will effect transformation in every facet of our business activities to make IBI 18 a rapid growth phase. With new drug candidates that can offer significant benefits to patients, such as emicizumab, atezolizumab, nemolizumab and SA237, as well as the steady generation of projects by CPR and the creation of middle molecule drugs, Chugai's path to future growth is already in sight.