3.An unprecedented effort to deliver medicines to more patients sooner
nemolizumab (CIM331)
Chugai's First Foray into the Dermatological Field
Based on a New Mode of Action
Nemolizumab is unique among Chugai's pipeline projects. An anti-IL-31 receptor A humanized monoclonal antibody targeting atopic dermatitis, it is Chugai's first foray into global development in the dermatological field.
Dr. Michiaki Tanaka was appointed global project leader for nemolizumab in September 2008. Originally a researcher in one of Chugai's laboratories, he expressed strong interest in IL-31 from the very beginning. He says he intuitively felt that “IL-31 was a very interesting cytokine because its major physiological function was to cause itching. Unlike other anti-cytokine antibodies, nemolizumab did not appear to suppress immunity. I thought it would be a medicine that could be used safely.”
With the start of the project, all team members began to learn about dermatology to sharpen their expertise, which gave them a renewed understanding of the significance of atopic dermatitis, a refractory chronic disease for which no effective new treatments had been launched in a long time. Severe pruritus can significantly reduce quality of life in various ways, such as by disrupting sleep, and the skin lesions caused by the itch-scratch cycle may adversely affect a patient’s physical appearance. Since many patients are children or teenagers, the condition may also interfere with studies. Because of these effects, the disease places a significant burden on patients and their families.
“There was an unmet medical need, and people’s desire for a new treatment was strong. It was an unknown area for us, but listening to the views of dermatologists deepened our recognition that this was a challenge we had to take on,” says Dr. Tanaka. Given that just about everything in the project was a first for the team, he set an unprecedented development policy by considering global studies from the very beginning.
An Unprecedented Development Policy
Enabled a Unique Phase I Clinical Trial
At almost the same time as they transitioned to development research (see page 78 for details), Dr. Tanaka’s team began to consult with physicians in Europe and the United States. There was little precedent for this at Chugai in early-stage projects, but the team set up an advisory panel made up of dermatology experts, and conducted market research not just in Japan, but globally. In addition, Dr. Tanaka decided to bring in patients as subjects for the phase I clinical trial in Japan. That decision was questioned inside the Company, as the standard practice until then had been to include patients from phase II of clinical development for indications other than cancer.
“Since the drug had a novel mode of action, of course it was very important to fully consider safety, but because the cytokine is specifically linked to itching, no side effects appeared in animal studies,” says Dr. Tanaka. In the course of internal discussions on consultation with regulatory authorities, the team received strong support from members of the Regulatory Affairs Department, who said that we should proceed since Chugai has nothing to lose by doing so. The whole nemolizumab project team then embarked on this great challenge.
“We were assuming out-licensing to third-party companies that specialize in the dermatological field,” says Dr. Tanaka, who was working out a strategy to keep development of nemolizumab, a potential breakthrough drug, on track. The phase I clinical trial, which started in September 2011, was conducted in a form that anticipated phase II. The trial was divided into three parts, with healthy Japanese adults in Part A, healthy Caucasian adults in Part B, and patients in Part C. Preliminary efficacy was demonstrated with a single dose. Based on that positive result, out-licensing activities were initiated.
This approach would eventually become common at Chugai, and became the basis of a new model for development in which early PoC is obtained in phase I.


Chugai's First Independent Phase II Multinational Study
The principal purpose of phase II clinical trials is to set the dosage and administration. Dr. Tanaka’s team presumed that if this new drug was to be marketed globally, health authorities in other countries would want to see clinical results in European and American subjects. In addition, authorities in Japan were also starting to push for greater participation in international clinical studies. So the team embarked on Chugai's first independent multinational study.
In Europe, it was necessary to file an application in each country in which a clinical trial was to be conducted, and clinical trial plans specific to each country were also required. This meant the team had to revise the clinical trial plan repeatedly, and the study period gradually became longer. Recalling the difficulty of conducting the study in three regions, Dr. Tanaka says, “Since it was all new to us, everything was challenging. There were major differences between countries and between medical facilities, and they were hard to predict at the time, so the study ended up taking eight months longer than we had planned.” Another impediment was that Chugai was not well known by local physicians, as it had no clinical trial experience in dermatology overseas.
However, because the blocking of IL-31 was then a new discovery in the dermatological field, doctors eventually expressed interest and the phase II multinational study got off to a smooth start. In Part A, the main therapeutic effect was confirmed after 12 weeks of observation, and in Part B, efficacy and safety were confirmed in administration for one year. Part B has ended, and the team has published a paper*7 and has delivered a conference presentation.*8
- *7
- The results from Part A were published in the New England Journal of Medicine Online on March 2, 2017 (EST).
- *8
- The results from Part B were presented at the annual meeting of the American Academy of Dermatology on March 4, 2017 (EST).
Bringing Nemolizumab to Patients Worldwide
by Out-Licensing to Partners
After the phase II multinational study, Chugai began to out-license nemolizumab to third-party companies, concluding license agreements with Galderma Pharma S.A. of Switzerland in July 2016 and Maruho Co., Ltd. of Japan in September 2016.
Galderma is a global pharmaceutical company with a strong presence in the dermatological field. Chugai and Galderma share many ideas on disease and medicines. After seeing the results of our phase II multinational study, Galderma evaluated nemolizumab as “an innovative biotherapy candidate with first-in-class potential,” which led to the agreement.
Galderma will continue with clinical development in countries outside Japan and Taiwan while Chugai manufactures and supplies the investigational drug.
Maruho has an impressive track record in Japan in the dermatological field. It expressed strong interest in nemolizumab from early on, and entered into the agreement because it shares Chugai's commitment to bringing this treatment to patients as soon as possible.


The Company’s Support Got Us through Many Turning Points
A Japanese phase II study of nemolizumab for pruritus in hemodialysis patients began in August 2015. Chugai plans to continue to conduct development and marketing in Japan for this indication.
This project notably features a series of firsts for Chugai. Dr. Tanaka recalls that behind them all was the strong enthusiasm and determination of the project team members, who were responsible for their respective divisions at the time. Every situation they faced was a turning point, but he says that they were able to get through each one thanks to the full support of the Company.
“These recent projects, including nemolizumab, SA237, emicizumab (ACE910) and Alecensa, were paving the way for Chugai to become a global top pharmaceutical company. While we were very fortunate with nemolizumab in many ways, such as the successful out-licensing to third-party companies, it created an opportunity for the whole company to move in a new direction, and I’m pleased that it also led to organizational transformation.
We project team members are all proud to have participated in this groundbreaking project,” Dr. Tanaka says emphatically. “I like it when everything is a first,” he adds with a smile, promising to continue innovating for the benefit of patients.
-
We have overcome many difficulties with enthusiasm and the devoted efforts of our team members.
Michiaki Tanaka, D.V.M., Ph.D.
Lifecycle Leader
Primary Lifecycle Management Dept.
Project & Lifecycle Management Unit
-
Nemolizumab is a new innovative biotherapy candidate in dermatology, developed as a clearly differentiated first-in-class solution for patients with moderate-to-severe atopic dermatitis.
Thibaud Portal
Vice President
Prescription Global Business Unit
Galderma S.A.
-
We will apply our development experience in the field of dermatology to bring groundbreaking medicines to patients as soon as possible.
Chieko Tanaka
Project Manager
Project Management Dept.
Maruho Co., Ltd.
Atopic Dermatitis and Nemolizumab
Atopic dermatitis (AD) is a complex multi-factorial disease combining two types of anomalies: alteration of the epidermal barrier and hypersensitivity to allergens. It is a chronic, pruriginous, inflammatory dermatitis manifesting itself in the form of flares. Pruritus in hemodialysis patients causes systemic severe itching unassociated with inflammation, and like atopic dermatitis, causes a significant reduction in patients’ quality of life.
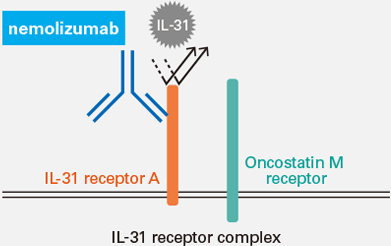
Chugai Research Institute for Molecular Medicine searched for new molecules that could potentially be candidate targets of new drugs, and in 1999 was first in the world to succeed in cloning the IL-31 receptor. The physiological function was not well understood at the time, but in 2004, it was found that IL-31 is the cytokine that causes itching. Chugai then created nemolizumab using its proprietary antibody engineering technology ACT-Ig.*3 There are expectations worldwide for nemolizumab as an innovative new treatment that suppresses the activity of IL-31 by competitively blocking binding with its receptor.
- *3
- Antibody half-life extending technology: pⅠ lowering engineering of the variable region improves antibody PK by repulsion with negatively-charged vascular endothelial cell surface.
■ Mode of Action of Nemolizumab (IL-31 Receptor A Antibody)
Galderma Pharma S.A. / Galderma S.A.
Headquartered in Switzerland, Galderma is a pharmaceutical company specializing in dermatology. Founded in 1981, the company now has a presence in 100 countries where it markets an extensive product portfolio to treat a range of dermatological conditions. The company partners with healthcare providers around the world to meet the skin health needs of people throughout their lifetime. Galderma is a leader in research and development of scientifically-defined, innovative medical solutions for the skin, hair and nails.
Maruho Co., Ltd.
Maruho is headquartered in Osaka and conducts research and development, manufacturing and commercialization of prescription pharmaceuticals. Founded in 1915, the company had 1,398 employees as of September, 30, 2016 and net sales of ¥70.1 billion for the fiscal year ended September 30, 2016. Pursuing its long-term corporate vision of “Excellence in Dermatology,” Maruho is striving to make significant contributions to the field.