Feature: 1. Quickly Making Innovative Medicines Available to Patients - Putting Patients First -
Individual Chugai Group employees involved in areas from discovery research to development, production engineering, and information provision talk about their daily work.
A Researcher’s Perspective


Aayam Lamichhane
Pharmaceutical Research Dept. 1, Research Division, Fuji Gotemba Research Labs
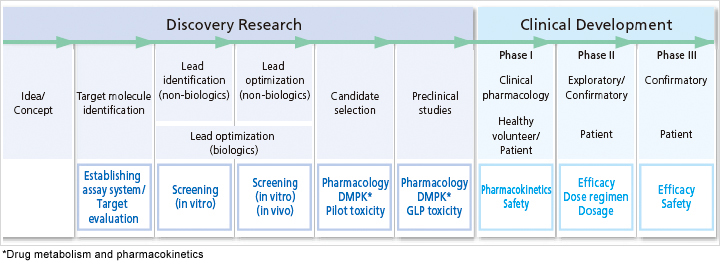
Since I joined Chugai four years ago, I’ve been involved in drug discovery research in the field of autoimmune diseases at the Fuji Gotemba Research Laboratories. Discovery research refers to the research leading up to the start of clinical trials that confirm a drug’s efficacy and safety in patients.
Identifying lead compounds accounts for 10 percent of the entire process and lead optimization for 40 percent. Tests using living cells and animal models to evaluate pharmacokinetics, safety and drug efficacy comprise the remaining 50 percent. I’m in charge of assessing drug efficacy throughout this process. Even when the results are good in animal models, unanticipated reactions can occur in the subsequent clinical trials. So thinking about how to design the candidate compound in order to improve its efficacy and disposition in patients is also an important job of researchers.
I think a distinguishing feature of Chugai’s drug discovery research is that it embodies the Company’s mission, which is to "create innovative medical products and services for the benefit of the medical community and human health around the world." The conventional approach to drug discovery is to start with an idea for a potential lead compound and work to commercialize it. At Chugai, however, we are committed to a patient-oriented approach, tying our technologies to the wishes of patients suffering from diseases and established ideas about the likely mechanisms behind those diseases to set the direction of research.
Therefore, Chugai actively collaborates with doctors who are familiar with patients and diseases, and with academics who are researching cutting-edge technologies.
Borrowing knowledge from outside the company can help to shorten research time and increase the success rate, allowing us to make new drugs available to patients sooner.
Drug Development Process
I was born in Nepal, and I decided to join Chugai because I wanted to help patients. The healthcare system and sanitary conditions in Nepal are not at the same level as Japan’s, so many people there suffer from illness. I was always thinking about what I could do for them. I had thought of pursuing a career as a medical doctor, but it occurred to me that if I became a researcher and created medicines, I could help people suffering from disease worldwide, including those in my own country. I chose to work at Chugai because it has a great environment for research.
The research project I’m working on now is progressing smoothly, and has started clinical trials. The drug under development targets a disease that affects a large number of patients. If we can make that drug available to those patients, it will be the greatest pleasure of my life, so I am watching its progress with anticipation.
A Developer’s Perspective


Keisuke Akada
Clinical Research Dept., Primary Research 2
Since I came to Chugai in 2005, I’ve been responsible for clinical trials in the field of liver disease, such as hepatitis B and C and compensated liver cirrhosis caused by hepatitis C virus (HCV). As it happened, clinical development of a treatment for compensated cirrhosis was starting right around the time I joined the company. I was involved in the entire clinical development process for about five years up until we put together the massive amount of clinical trial data and submitted it to the review body in October 2010.
At the planning stage of a clinical trial, we draw up a development design in consultation with outside experts. This design sets the criteria for selection of medical institutions to carry out the trials and the criteria for patient participation, the objective being to scientifically prove the efficacy and safety of the drug and get it to patients as soon as we can. The development design is incorporated into the clinical trial protocol. How can we shorten the duration of the clinical trial? How can we deliver the drug to patients faster? These questions have a significant impact on the development design, and a lot of energy goes into addressing them when drawing up the design, because that’s where our ingenuity comes into play.
An investigational new drug for compensated liver cirrhosis caused by HCV is now under review. Until it is approved, we need to carefully respond to each inquiry from and document inspection by the review body and to each study at the medical institutions conducting the clinical trial. After the one- to two-year review period, the safety and efficacy of the drug I was involved in developing may be recognized and at last it can be made available to patients. The joy this will bring me, I wouldn’t trade for anything.
At the medical institution where I was in charge of clinical trials for compensated liver cirrhosis caused by HCV, a benefit was seen in the patients taking the drug, and the doctors thanked me. I was really happy with the results.
An investigational new drug we are developing for hepatitis B is also one of the drugs requested for early approval in the "7-Year Strategy for Hepatitis Research" produced by hepatitis research specialists in Japan based on feedback from patient groups and many others concerned with the disease. We are keenly aware of the high expectations of patients and healthcare professionals, and that renews our resolve.
We should contribute to human health worldwide by quickly bringing best-inclass drugs and breakthrough drugs with novel mechanisms of action to patients in Japan and, by extension, to patients around the world. We apply ourselves to development on a daily basis with that thought in mind.
A Production Engineer’s Perspective


Yoshihiro Tomojiri
Production Engineering Dept. (Formulation Technology) Pharmaceutical Technology Div.
Over the last ten years, I’ve been involved in designing the packaging for Actemra® and other products and launching the packaging line at the Utsunomiya plant. Currently I’m in charge of package design for solid-form drugs at the Fujieda plant.
The important functions of pharmaceutical packaging are to preserve the quality of the medicine until it gets to the patient and to ensure its proper use. Therefore, the package must not only protect the medicine inside but also make it easy to use, without mistakes. We also have to design it from a total perspective that takes into account considerations such as environmental friendliness and stable, efficient manufacture. We must be able to envision the handling of the medicine from the time it leaves the plant until it is actually used. In addition to giving due attention to shock absorbency to avoid breakage during distribution, we focus on ease of use and safety. Since tablets are used by the patients themselves, while in many cases an injection is administered by a nurse or other practitioner, we think about where and by whom the product will be used for each formulation and indication.
Actemra® packaging, for example, has a compact design because the medicine is stored in a refrigerator at the healthcare facility. However, to increase breakage resistance if the box is accidentally dropped when removing it from the refrigerator, we have devised dividers that suspend the individual containers a little inside the box. For press-through packages (PTP) used for tablets and capsules, we are researching human behavioral traits to develop PTP sheets that are easy to handle and will not be accidentally ingested.
As a group of engineers, we constantly think about the patients who will use the medicine, honing our technological capabilities every day with the belief that it is important to ensure the safe, proper use of high-quality pharmaceuticals and continuously provide a stable supply.
In new product development projects, I also have the opportunity to visit hospitals as a package designer and hear various opinions. What I’ve learned from listening to people who actually use the medicines is that they have very detailed requests, and also that their needs change as their environment changes. I’m realizing that in the project I’m currently working on, which has been quite an education.
To respond quickly to the ever-changing-needs of the medical community, we are now collecting and analyzing information on needs throughout the organization, and are creating a plan for strengthening mechanisms that reflects those needs in development policies. We designers provide technology and value to the market in the form of packaging, which is used by patients. Then we identify patients’ underlying needs and reflect them in new products. I believe that fulfilling this responsibility will lead to better pharmaceutical packaging.
An MR’s Perspective


Mitsuka Saito
Yokohama Branch, Primary Unit, Yokohama Sales Group, Yokohama General Sect. 1
In the five years that I’ve been with Chugai, I’ve been responsible for clinics and hospitals as an MR, and worked closely with the doctors at those facilities. Since October 2010, I’ve been assigned to university hospitals and have been diligently providing information and making prescription suggestions, with a focus on Actemra® for rheumatoid arthritis and other indications,and Pegasys® for chronic hepatitis C.
The job of MRs is directly linked to the lives of patients, so we have to make a string of choices: What is the best treatment for this patient? Which medicine will work best for that treatment? Of course, we want them to use our own company’s medicine. However, we make suggestions with the patient foremost in mind, based on laboratory test values and other data. After confirming the patient’s condition, I may say to the doctor, "Given the potential risks, what about this medicine?" or "It might be good to use these medicines in combination."
Chugai’s strength is that it has innovative drugs for specific diseases. Actemra® for rheumatoid arthritis and other indications is one example. The fact that the drug is useful for patients makes it especially important to properly communicate safety and other information. University hospitals also have the function of conveying information to regular hospitals. For that reason, Chugai’s MRs also properly share information with affiliated hospitals and partner hospitals, thereby contributing to better overall quality of care.
I think that in the future it will be necessary to create a cycle in which I can push the Company to craft measures that address the problems and needs that I identify at hospitals or clinics. I want to help make good medicines available to patients by building relationships of trust with doctors so they see MRs as partners in healthcare.
When I was a newcomer at Chugai, the Company had just launched a new medicine for breast cancer, and I was introducing that anti-cancer agent to a doctor. The doctor came right out and told me his concern — that even though he knew the medicine had shown good results in clinical trials overseas, he wondered how safe it would be for Japanese patients. But he decided to use it, partly because the patient was willing. After he started administering the drug, I visited the doctor on each of the patient’s appointment dates, asking him about side effects and the lab test results. I would quickly check on anything he seemed concerned about and report back to him. After about three months, he told me, "The patient is really happy with this drug because it is comfortable to use and hasn’t caused any hair loss or fatigue." I was so glad when I heard that. The doctor even showed me a photo of him with the patient that was taken as a keepsake. The smile on the patient’s face is still imprinted in my mind.
An MR’s Perspective


Kazuteru Ono
Tokyo Branch 1, Oncology Unit, Tokyo-Chiba Sales Group, Tokyo Oncology Sect. 3
When I graduated from university, I joined another pharmaceutical company, but I was particularly drawn to the field of oncology. I thought I’d like to work at Chugai, which has a strong lineup of oncology drugs, and joined the Company six years ago. Since then, as an MR I’ve been in charge of cancer hospitals. Right now I mainly deal with treatments for breast cancer and lung cancer. Since I joined Chugai in 2005, three new anticancer agents — Avastin®, Femara® and Tarceva® — have been launched. Including existing drugs with additional indications, we’ve been able to offer new medicines every year.
Chugai now has an extensive lineup of medicines covering four of the five major cancer types, and most of them are listed in treatment guidelines as first-line treatments. That makes it easy to match our proposals with what doctors are requesting. I am proud to have gained enough trust that doctors feel they can count on Chugai to provide sound advice about cancer treatment.
On the other hand, with so many medicines available and the high level of need at care facilities, I sometimes wonder how I can conduct detailed information activities for each doctor. It’s a good problem to have.
Today Chugai continues to innovate for patients as it pushes toward its target of becoming a top Japanese pharmaceutical company. MRs work at the point of care and play a pivotal role as a bridge to appropriate medicines by listening to the frank opinions of doctors who are familiar with the patients. I’m proud to be able to do that. I want to meet sales targets, of course, but I’m also working to enhance my abilities as an MR by acquiring broad scientific knowledge and practicing thorough compliance. I aim to be the best in everything I do.
I was approached for advice by a doctor who was unsure about how to treat a particular patient. The patient was elderly, and had no desire to be treated with strong medicine because she wanted to enjoy the rest of her life. I consulted with my superior and more experienced MRs, and looked up similar cases. After much agonizing, I decided to suggest another company’s product rather than a Chugai product. I cannot say I am truly acting for patient-centered healthcare if I recommend our company’s medicine when I know it may not be right for the patient. Chugai’s policy of putting patients first permeates the Company, and it’s because of that policy that I am able to put aside my conflict as a sales representative and make decisions like this.
I’m delighted that I was able to propose a solution for the patient and doctor in this instance. By being honest with doctors, I can build stronger relationships of trust, and helping them to gain a deep understanding of Chugai’s products makes my work doubly enjoyable and satisfying.
