Intellectual Property
The Role of Intellectual Property Activities at Chugai
The Role of IP Activities in the Pharmaceutical Business
R&D-oriented pharmaceutical companies, such as Chugai, operate on a business model where they recoup vast R&D investments during a period of market exclusivity granted by intellectual property (IP) rights, primarily patents. The profits generated from these new drugs are therefore the driving force for future drug discovery.
However, when the term of the basic patent, which is the basis of the product, expires, sales drop sharply due to the market entry of generic drugs or biosimilars. This phenomenon is called “patent cliff” and is a major management issue common to R&D-oriented pharmaceutical companies.
To address this challenge, Chugai is promoting strategic IP activities that go beyond the mere protection of inventions to maximize product value and support the company’s sustainable growth. To maintain product competitiveness and mitigate a sharp decline in sales even after the expiration of the basic patent, the company is building a robust patent portfolio that protects products in a multifaceted manner by acquiring not only basic patents but also lifecycle management patents for related technologies that add value to the product.
Increasing complexity of drug discovery and changing role of the IP organization
Diversification of modalities and solutions
In recent years, new modalities such as gene therapy and regenerative medicine have emerged, making drug development much more advanced and complex than in the past. In addition, the solutions required for healthcare are expanding beyond therapeutics.
This diversification has also led to changes in the way IP is conducted. Since many patents and patent owners are involved in a single product, peripheral service, or solution, it has become difficult for a single company to exclusively control all related technologies.
Reflecting the changes in the business environment, Chugai’s IP activities are also evolving. To supplement its in-house capabilities, the company is emphasizing the concept of co-creation-which includes collaborations with other companies and open innovation to bring in external technologies-and is establishing and implementing new IP activity policies to facilitate partnerships with external entities.

The Role of the IP Organization at Chugai
Creating and utilizing intellectual property rights, as well as securing freedom to operate by respecting the IP rights of other companies, is crucial for maximizing business value.
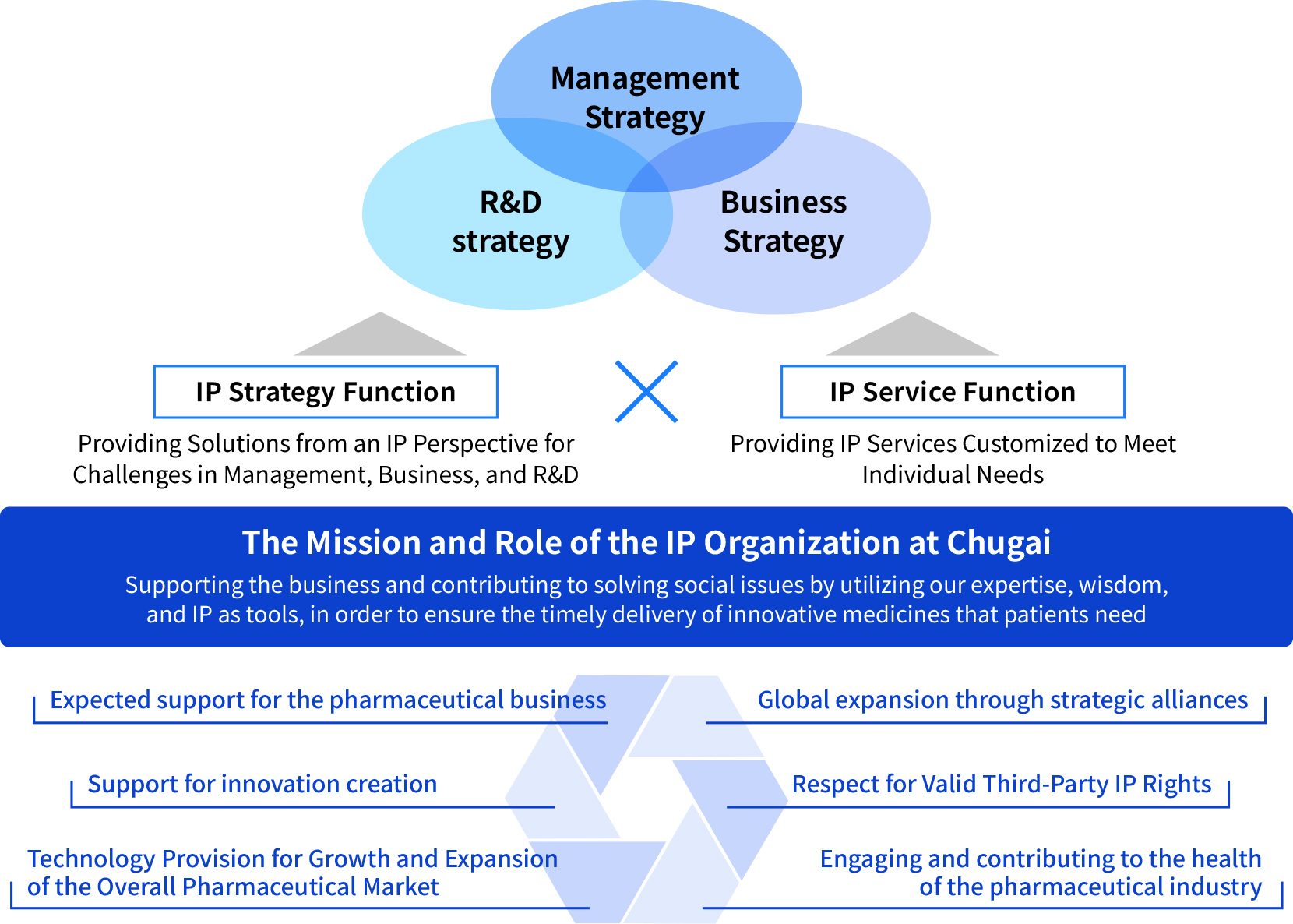
To support our growth scenarios with “strategic IP” that is closely aligned with the three layers of management, business, and R&D, our IP organization has defined six basic roles, centered on the two functions of the IP strategy function and the IP service function. Furthermore, we defines its mission as supporting the business and contributing to solving social issues by utilizing our expertise, wisdom, and IP as tools, in order to ensure the timely delivery of innovative medicines that patients need.
IP Strategy Function
We propose approaches and solutions from an IP perspective to address the challenges faced by Management, Business, and R&D. We continuously strengthen our role as an IP organization that contributes to corporate strategy and challenge ourselves to engage in problem-solving initiatives.
IP Service Function
We provide Custom-made IP services to meet individual needs.
Direction of Chugai IP Activities
Chugai’s IP activity policy is centered on the two wheels of: an “IP service function” that provides custom-made support for the individual needs of our business and R&D divisions, and an “IP strategy function” that supports the resolution of management issues from an IP perspective.
Through these functions, the IP organization aims to continue being a strategic IP organization that contributes to enhancing corporate value. As a compass to realize this goal, the organization has established the following five basic policies.
These policies are centered on IP portfolio management aimed at maximizing the value of our pharmaceutical business and creating innovation. We are further strengthening the strategic development and optimization of this portfolio and promoting our daily IP activities.

IP Activities to Support the Competitive Advantage of Our Products
Protection of Platform Technologies
Chugai is strengthening IP activities to support our “Technology-Driven” drug discovery capabilities, which are based on unique technology and science, and the competitiveness of our products. The total number of patents we hold is steadily increasing. While maintaining a strong patent network in the antibody drug field, which is one of our strengths, we are also building a patent network in macrocyclic peptide drugs field.
In the antibody & biotechnology field, in addition to the patent portfolio for our products and development pipeline, patents protecting our innovative drug discovery platforms-such as unique antibody engineering technologies account for approximately 20%. Some of these technologies are also licensed out to other companies.
In the field of the macrocyclic peptide, a new growth area, patents related to our unique drug discovery platform technologies constitute the majority. This includes a variety of technologies essential for drug discovery R&D in the macrocyclic peptide, and we are expanding our patent network year by year to support our R&D activities.
![Patent Family Ratio by Modalities: [Fy2020] Antibodies & Biotechnology - 75%, Mid-size molecule - 6%, Low molecule - 15%, others - 4% [FY2024] Antibodies & Biotechnology - 66%, Mid-size molecule - 14%, Low molecule - 13%, others - 7% / Antibodies & Biotechnology [FY2024] Ab-Tech - 19%, Project/Product Related, Formulation, Manufacturing, Etc. - 81% Our unique group of innovative antibody engineering technologies is protected by patents. These patents form a key foundation that supports our technology-driven R&D. / Mid-size molecule [FY2024] Microcyclic peptide Platforms Technology - 77%, Project related - 23% We are building a strong patent portfolio to support our unique drug discovery platform technology related to macrocyclic peptide platforms. This includes technologies essential for the drug discovery process, such as peptide screening and chemical synthesis.](/english/innovation/rd/intellectual/images/pic_intellectual05.jpg?202604)
For more information
Protection and Maximization of the Value of Our Products
To maximize the value of our products, we are strengthening our efforts to build a multi-layered and robust patent portfolio. We construct a robust patent network by combining platform technology patents, composition of matter patents that form the core of our products, and life cycle patents, all in anticipation of potential disputes with third parties.
Simply building a patent portfolio is not enough to maintain a product’s competitive advantage over the long term. We place particular emphasis on the process of building a strong and optimally sized patent portfolio with a perspective on the entire product life cycle, and strategically leveraging it-including through legitimate enforcement of rights and licensing-to maintain and enhance product value. These IP activities allow us to maintain product competitiveness by preparing for or responding in a timely and appropriate manner to changes in the market environment, the competitive landscape, and challenges from all angles. Maximizing product value through such efforts serves as the source of investment for creating future innovations.
Therefore, our patent strategy is formulated in close collaboration with our R&D and business divisions. Especially at important milestones, such as selecting countries for filing or transitioning between R&D phases, we balance the optimization of our patent portfolio with the maximization of product value by closely aligning our IP strategy with the strategies and progress of each division.
Another important aspect of patent portfolio management is ensuring Freedom to Operate (FTO). To ensure that innovative medicines can be delivered to patients, it is essential not only to solidify our own rights but also to respect the valid third-party IP rights and minimize potential IP risks.
Securing FTO to Minimize Potential Risks
We conduct high-precision third-party patent searches and perform IP risk assessments for FTO at each phase transition-from the early stages of R&D for our products through to the marketing approval application-in order to minimize potential risks. For example, we conduct a more in-depth assessment of IP risks at critical business milestones, such as upon completion of the Proof of Concept (PoC) for efficacy and safety, a point at which the probability of technical success increases.
IP Risk management and Respect for Valid Third-Party IP Rights
To fulfill our mission of continuously creating and delivering innovative medicines to patients, we place great importance on securing our freedom to operate.
As our fundamental approach, we have a respect for IP, treating valid third-party IP rights with the same respect as our own.
Therefore, from R&D stage, we continuously monitor third-party patents and other IPs, and constantly assess any risks that could impact our business. As a result, if we determine that a patent has the potential to unreasonably restrict our freedom to operate, we will take necessary actions after carefully examining its validity. Such actions may include, where appropriate, filing an opposition or a request for an invalidation trial against the third-party patent.
Global Expansion through our Strategic Alliance with Roche
We work closely with Roche as One Team in all aspects of IP, from contract negotiations to securing and leveraging rights, to maximize the value of our strategic alliance:
-

Collaborating in patent portfolio management to maximize LOE* in each region (supporting in- and out-licensing activities)
- *Loss of Exclusivity
-

Actions to IP-related disputes and litigation
-

Sharing frameworks for IP operations between the two companies, such as criteria for patent/trademark filing countries, IP-related databases, use of generative Al, copyright, and OSS
-

Implementation of a talent exchange program between Chugai Pharmaceutical and a Roche Group specializing in IP talent.
For more information
Acceleration of Innovation Creation
Maximizing co-creation synergies through open innovation
Chugai is going to further expand open innovation in its growth strategy toward 2030, “TOP I 2030” (FY2021-FY2030).
To ensure the success of such co-creation, flexible contract and license design tailored to the objectives of the collaboration and the specific partner is essential. Consequently, it is required not only to acquire and manage IP rights, but also to design and support strategic partnerships that maximize the synergies from co-creation.
To this end, the IP organization is collaborating closely with the R&D and business divisions to implement flexible IP utilization that goes beyond simply protecting its own technologies.
For more information
Accelerating Innovation with IP Intelligence
The role of IP is not limited to merely protecting the technologies we have created. For exploring new research themes and developing new business areas, technical information from around the world, such as patents and scientific papers, is a veritable treasure trove. We analyze this vast amount of information rapidly and from multiple perspectives by combining several of the latest IP information analysis tools equipped with Generative AI and AI functions.
We are strengthening our IP intelligence, the strategic utilization of IP information. Through close collaboration between the IP organization, the R&D division, the business division, and external partners, we derive optimal problem-solving solutions and accelerate the creation of innovation.
For this purpose, we have members of our IP department stationed at our major research sites (*LSP-Yokohama, IFReC, and CPR). Through daily communication between researchers and IP department members, we promptly identify the early signs of inventions and collaboratively develop strategies to maximize their value. Our IP organization, through its on-site-oriented system, promotes the creation of high-quality inventions and accelerates the speedy and strategic acquisition of rights.
- (*)Closely working with members of the IP department
Chugai Life Science Park Yokohama, Osaka University Immunology Frontier Research Center (IFReC), Chugai Pharmabody Research Pte. Ltd.
Promoting Innovation through the Employee Invention Incentive System
We have revised our employee invention incentive system to further motivate employees who take on the challenge of innovation, for example by establishing a new incentive category for “innovative technologies” and enabling earlier payment of rewards for patents that protect our products. Through these initiatives, we promote “Technology-Driven” drug discovery based on Chugai’s unique technologies, while also contributing to the achievement of the high goals set forth in our growth strategy, “TOP I 2030”.
Governance System with High Transparency
IP is a top management priority. We have established a highly transparent governance system to ensure that our IP strategy consistently aligns with and supports the execution of our business strategy.
Specifically, we regularly report to the Board of Directors on our IP activities (including our IP strategy policy, the status of our IP portfolio, important litigations and disputes, and the status of cases that could significantly impact our business), and conduct these activities under the supervision of the Board.
Contributing to the growth and expansion of the pharmaceutical industry as a whole and upholding its integrity.
Utilization and Licensing-out of Chugai’s Unique Antibody Engineering Technologies
Chugai’s unique antibody engineering technologies support our drug discovery efforts as a platform technology applied to our in-house developed products. Some of these have also been utilized in several of our commercialized products, and serve as the source of their competitive advantage.
Furthermore, with the aim of contributing to global healthcare and people’s health, we do not exclusively use these superior unique technologies for ourselves. Our IP organization, R&D and business divisions collaborate closely to help our out-licensing partners utilize the licensed technology smoothly and to its fullest potential.
For more information
Chugai’s position on Intellectual Property and Access to Health
The Mission set forth in the Mission Statement of the Chugai Group is to “Dedicate ourselves to adding value by creating and delivering innovative products and services for the medical community and human health around the world”.
The Core Values are our most important standards of judgment that will be applied while engaging in activities that support our realization of the Mission and we will operate our business in accordance with them. The Chugai Group will contribute to the realization of a sustainable society by solving social issues through the creation of innovation and efforts toward respect for the environment, human rights, and more.
We believe that robust Intellectual Property (IP) systems stimulate such innovation and solving social issues for the benefit of society as a whole. At the same time, we also believe that it is important to carry out our IP activities in consideration of the public interest. Based on those principles, we established our position on Intellectual Property and Access to Health in the Executive Committee.
The full text of Chugai’s position on Intellectual Property and Access to Health