Chugai News Releases are issued to provide stakeholders with the most up-to-date information related to our company. In some instances, information on products or drug candidates under development may be included, but this is intended for members of the media, shareholders, and investors. The information is not intended for promotional or advertising purposes, or as medical advice, etc.
Jul 21, 2022
- Corporate
Chugai Announces 2022 Half Year Results
- Record-high core revenues and core operating profit in the first half at ¥504.3 billion (+29.2%) and ¥201.4 billion (+21.5%), respectively
- Steady growth in revenues and profits in the second quarter driven by the contribution of new products following the strong first quarter
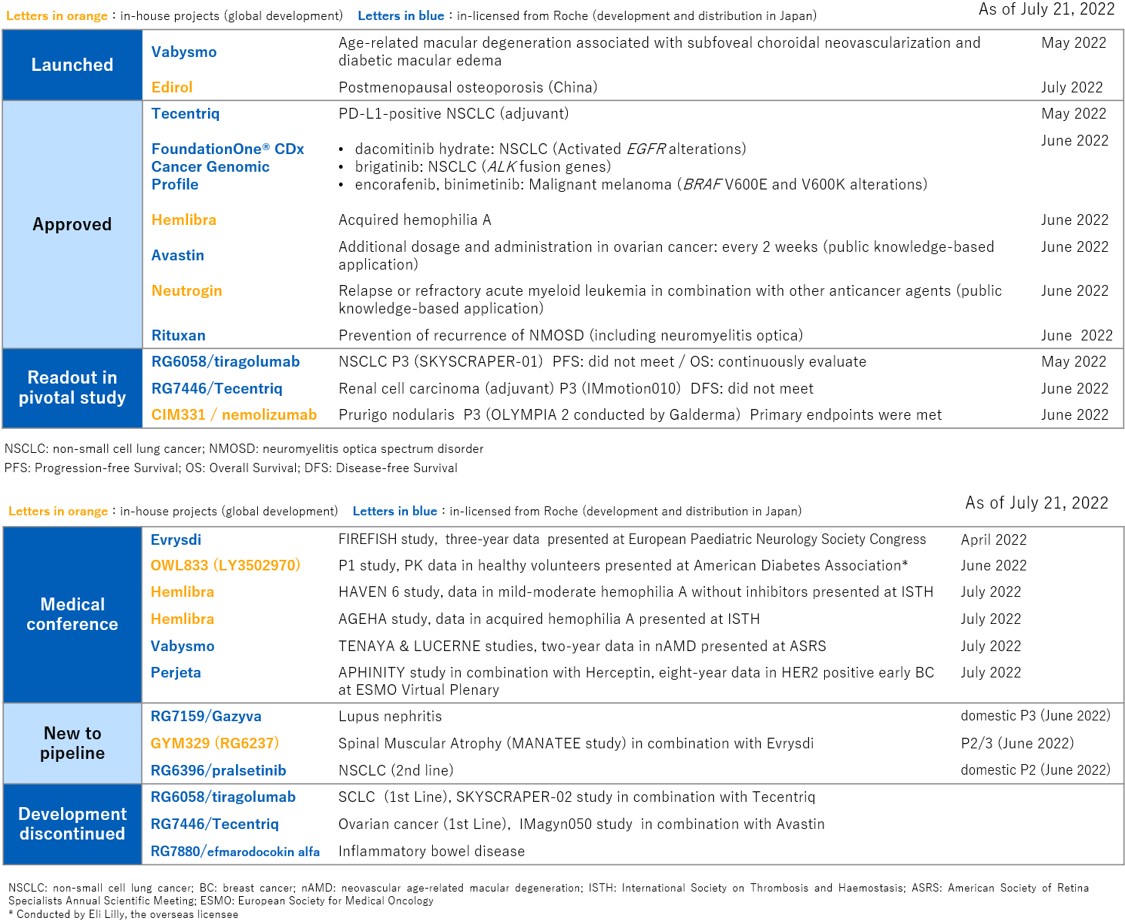
- Sound progress in R&D, including nemolizumab achieving the primary endpoints in a phase III study for prurigo nodularis and approval for additional indications of Hemlibra and Tecentriq
“Chugai achieved record-high revenues and profits in the first half of 2022 as our strong first-quarter performance was followed by steady increases in both revenues and profits during the second quarter. Results were driven by growing market penetration of new products Enspryng®, Polivy®, Evrysdi®, and the new ophthalmic drug, Vabysmo®, launched in May 2022. On the R&D side, nemolizumab, an investigational antibody originated by Chugai, achieved the primary endpoints in the first phase III clinical trial for prurigo nodularis, as announced by our overseas partner Galderma. The results demonstrated that the drug candidate may potentially provide new value to patients with a disease for which no approved drugs are available. Hemlibra® and Tecentriq® each obtained regulatory approval for an additional indication, offering a new treatment option for the patients. Hemlibra was approved for acquired hemophilia A, and Tecentriq became the first cancer immunotherapy in Japan to receive approval for adjuvant therapy in non-small cell lung cancer. We will continue to focus all our efforts on innovation to contribute to patients waiting for new treatments,” said Dr. Osamu Okuda, Chugai’s President and CEO.
Half Year Financial Results (Core results, January to June 2022)
Chugai reported record-high financial results for the half year 2022 as revenues increased by approximately 30% and operating profit increased by approximately 20% over the same period last year.
An increase in domestic and overseas sales outweighed a decrease in royalties and other operating income, resulting in a 30% increase in total revenues. Sales increased by approximately 50%, of which domestic sales increased by approximately 30%, and overseas sales increased by approximately 80%. In domestic sales, the Oncology field reported flat growth while the Specialty field increased by approximately 90%. In the Oncology field, contributions of the new product Polivy and an increase in Kadcyla® sales offset declines in mature products, including Avastin® and Herceptin®, due to NHI drug price revisions and biosimilars. In the Specialty field, renamed from the Primary field following the organizational change in July, there were contributions from the supply of Ronapreve to the government in the first quarter, as well as an approximately 20% increase in sales of the mainstay product Hemlibra, and steady market penetration of new products, Enspryng, Evrysdi, and Vabysmo, which was launched in May of this year. The increase in overseas sales was driven by a 2.7-times increase of Hemlibra owing to the full-scale export to Roche at regular shipping price and a 70% increase in Actemra®, which obtained emergency use authorization and regulatory approval for severe COVID-19 in the U.S. and Europe, respectively, since last June. On the other hand, royalties and other operating income decreased by approximately 40%, mainly due to a significant decrease in royalty income related to the initial shipments of Hemlibra.
Cost to sales ratio rose by 2.7 percentage points year-on-year to 42.8%, mainly due to a change in the product mix. Operating expenses grew by 6.5% as both marketing and distribution, and research and development expenses increased, while general and administration expenses decreased. Marketing and distribution expenses increased mainly due to foreign exchange effects. Research and development expenses increased due to the progress of projects under development and foreign exchange effects. General and administrative expenses decreased primarily due to gain on sales of property, plant and equipment, despite increases in the enterprise tax and various expenses. As a result, Core operating profit totaled ¥201.4 billion (+21.5%).
Quarterly Financial Results (Core results, April to June 2022)
Revenues for the second quarter (Apr-Jun) increased by 6.4% year-on-year, as an increase in sales driven by overseas sales exceeded the significant decline in royalties and other operating income. Sales increased by approximately 20%, of which domestic sales increased by approximately 3%, and overseas sales increased by approximately 50%. The increase in domestic sales was driven by a double-digit growth in the Specialty field, which exceeded a decline in the Oncology field. The supply of Ronapreve to the government, which was the main reason for the 70% year-on-year increase in domestic sales in the first quarter, was not reported in the second quarter. Overseas sales of Hemlibra and Actemra increased both by more than 80%. The cost to sales ratio was almost at the same level as the same period of last year, while it improved compared to the first quarter of this year by approximately 9 percentage points due to a higher proportion of in-house products in total sales. Despite reporting double-digit growth in the first quarter, operating expenses in the second quarter were comparable to the same period of last year, mainly due to the reporting of gain on sales of property, plant and equipment in general and administrative expenses. As a result, Core operating profit for the second quarter increased by approximately 2%.
R&D activities
The Company also made good progress in research and development. Among in-house products, nemolizumab, an investigational antibody with a novel mode of action, achieved the primary endpoints in one of the two ongoing phase III clinical trials for prurigo nodularis, as announced by our overseas partner Galderma in June. Hemlibra, an anti-coagulation factor IXa/X humanized bispecific monoclonal antibody, was approved for the additional indication for acquired hemophilia A based on a domestic phase III AGEHA study in June as well. Furthermore, a clinical trial investigating GYM329 in combination with Evrysdi was initiated for spinal muscular atrophy. As for in-licensed products from Roche, Tecentriq, a cancer immune checkpoint inhibitor, was approved in May as the first cancer immunotherapy in Japan for adjuvant treatment of non-small cell lung cancer.
2022 half year results (January to June)
| Billion JPY | 2022 Jan - Jun | 2021 Jan - Jun | % change |
|---|---|---|---|
| Core results | |||
| Revenues | 504.3 | 390.2 | +29.2% |
| Sales | 452.8 | 304.1 | +48.9% |
| Royalties and other operating income | 51.4 | 86.1 | -40.3% |
| Operating profit | 201.4 | 165.8 | +21.5% |
| Net income | 144.7 | 121.7 | +18.9% |
| IFRS results | |||
| Revenues | 596.2 | 390.2 | +52.8% |
| Operating profit | 286.9 | 160.7 | +78.5% |
| Net income | 204.2 | 118.1 | +72.9% |
Sales breakdown
| Billion JPY | 2022 Jan - Jun | 2021 Jan - Jun | % change |
|---|---|---|---|
| Sales | 452.8 | 304.1 | +48.9% |
| Domestic sales | 273.8 | 203.4 | +34.6% |
| Oncology | 123.0 | 124.1 | -0.9% |
| Specialty | 150.9 | 79.3 | +90.3% |
| Overseas sales | 179.0 | 100.7 | +77.8% |
Progress in R&D activities from Apr 26th, 2022 to Jul 21st, 2022
About Core results
Chugai discloses its results on a Core basis from 2013 in conjunction with its decision to apply IFRS. Core results are the results after adjusting non-Core items to IFRS results. Chugai's recognition of non-recurring items may differ from that of Roche due to the difference in the scale of operations, the scope of business and other factors. Core results are used by Chugai as an internal performance indicator, for explaining the underlying business performance both internally and externally, and as the basis for payment-by-results such as a return to shareholders.
Trademarks used or mentioned in this release are protected by law.Contact:
- For Media
- Chugai Pharmaceutical Co., Ltd.
- Media Relations Group, Corporate Communications Dept.,
- Tomoko Shimizu
- Tel: +81-3-3273-0881
- E-mail: pr@chugai-pharm.co.jp
- For Investors
- Chugai Pharmaceutical Co., Ltd.
- Investor Relations Group, Corporate Communications Dept.,
- Takayuki Sakurai
- Tel: +81-3-3273-0554
- E-mail: ir@chugai-pharm.co.jp