Chugai News Releases are issued to provide stakeholders with the most up-to-date information related to our company. In some instances, information on products or drug candidates under development may be included, but this is intended for members of the media, shareholders, and investors. The information is not intended for promotional or advertising purposes, or as medical advice, etc.
Feb 01, 2024
- Corporate
Chugai Announces 2023 Full Year Results and Forecasts for 2024
- Core revenue, core operating profit and core net income for the fiscal year 2023 at ¥1,111.4 billion (-4.8%), ¥450.7 billion (-0.2%) and ¥333.6 (+5.0%) respectively
- Steady progress in R&D activities for both early and late-stage development, including initiation of clinical trials of several in-house projects applying Chugai’s innovative proprietary antibody engineering technologies. Also with mid-size molecules, the blood transfer after oral administration of LUNA18 was confirmed and subsequent projects are substantial. Development of crovalimab and Alecensa also made progress
- Planned 2023 year-end dividends are ¥40 per share (annual dividends for the fiscal year: ¥80 per share)
- Core revenues and core operating profit in 2024 are expected to be ¥1,070.0 billion (-3.7%) and ¥460.0 billion (+2.1%), respectively
TOKYO, February 1, 2024 -- Chugai Pharmaceutical Co., Ltd. (TOKYO: 4519) announced its consolidated financial results for the fiscal year ended December 31, 2023, and forecasts for the fiscal year ending December 31, 2024.
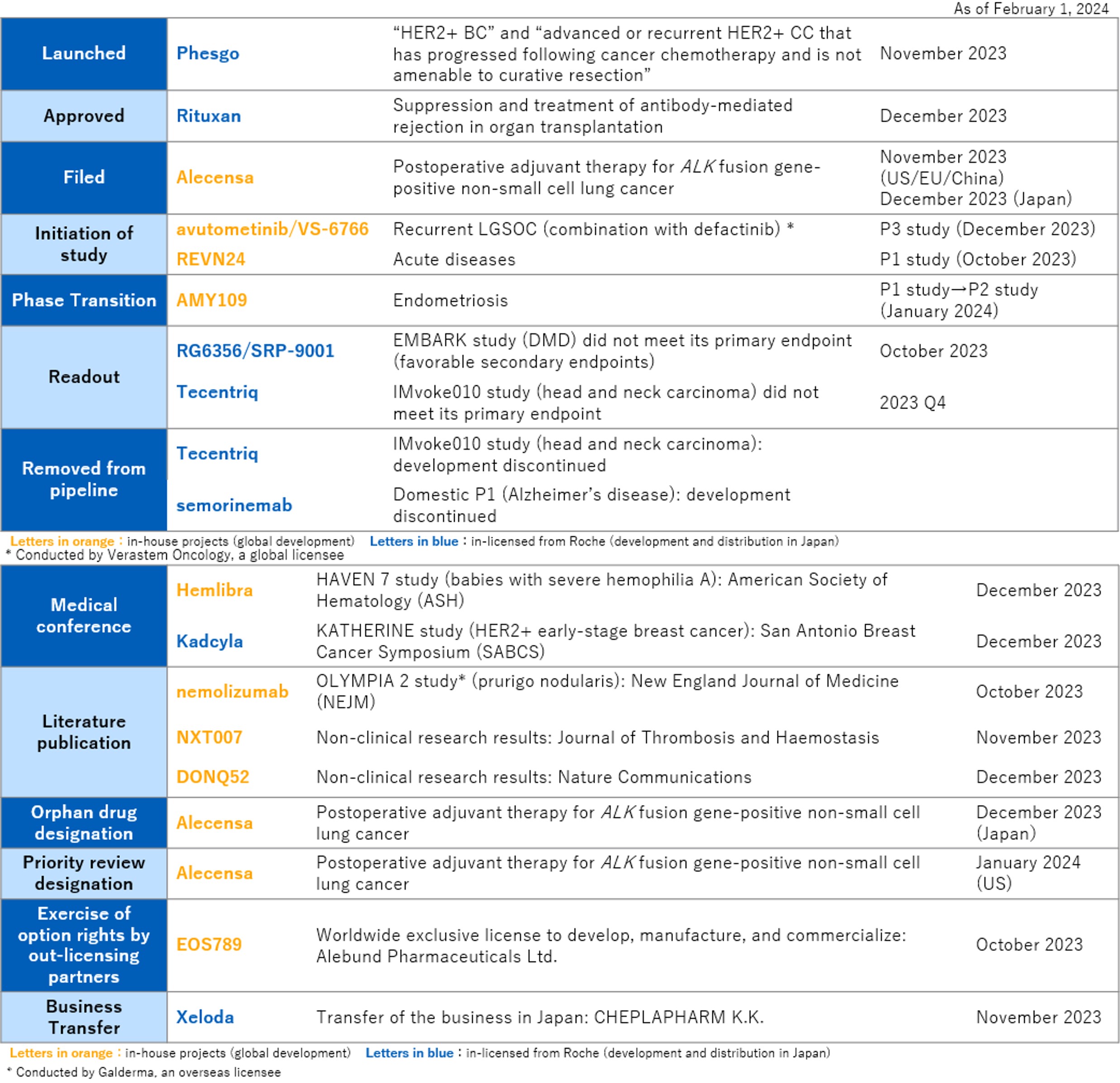
“In 2023, the third year of Chugai's growth strategy TOP I 2030, revenues decreased due to the decrease in the supply of Ronapreve® for COVID-19 treatment to the government. However, for the second consecutive year, we were able to achieve revenues of more than ¥1 trillion, continuing from the previous year and core net income increased. In November, we launched Phesgo®, a subcutaneous combination of Perjeta® and Herceptin®, the standard treatment for HER2-positive breast cancer, to reduce infusion time and help improve the daily lives of patients. In R&D, a number of in-house projects that apply our innovative proprietary antibody engineering technologies have entered the clinical development stage, and we have more than 10 in-house projects in the early development stage. For mid-size molecules, which we expect as the pillar for the third drug discovery modality, we have confirmed the important concept of blood transfer after oral administration in the clinical trial of LUNA18, and we continue to see numerous subsequent projects. In late stage development, we are making steady progress, with simultaneous applications in Japan, the United States and Europe for the treatment of paroxysmal nocturnal hemoglobinuria (PNH). In 2024, we will continue to take on unprecedented challenges and pursue innovation to address unmet medical needs with global first-class drug discovery capabilities,” said Dr. Osamu Okuda, Chugai’s President and CEO.
<Full year Core results for 2023>
Chugai reported that revenue for the fiscal year ended December 2023 totaled ¥1,111.4 billion (-¥56.4 billion, - 4.8%), exceeding ¥1 trillion for the second consecutive year.
Domestic sales were ¥558.0 billion (-¥96.7 billion, -14.8%). In the oncology field, the sales were comparable to the same period of last year due to the contribution of steady market penetration of new product Polivy®, a treatment for malignant lymphoma, and the growth of mainstay product Tecentriq®, an immune checkpoint inhibitor, despite the impact of biosimilars and NHI drug price revisions on mature products such as Avastin® and Herceptin. In the Specialty field, the sales decreased by approximately 25% compared with the same period in the previous fiscal year due to a substantial decrease in the supply of Ronapreve for COVID-19 treatment to the government, while new products including Vabysmo® in the ophthalmology area, mainstay products such as Hemlibra® for hemophilia and Enspryng® for neurology contributed to sales growth. Overseas sales were ¥416.5 billion (+¥31.9 billion, +8.3%), driven by a substantial increase in exports of Hemlibra and Alecensa®. Other revenue increased by nearly 10% mainly due to increase in lump-sum income, etc., in addition to the increase in income related to Hemlibra. Revenue on IFRS basis, including Non-Core items, decreased due to the non-recurrence of upfront payment income from the settlement agreement with Alexion Pharmaceuticals, Inc., in the previous year.
Cost to sales ratio improved by 3.4% points year-on-year to 42.3%, mainly due to a change in the product mix, despite the negative impact of foreign exchange. Research and development expenses amounted to ¥162.8 billion (+13.3%) due to investments into drug discovery and early development, including the start of full-scale operation of Chugai Life Science Park Yokohama, and the progress of development projects. Also selling, general and administration expenses increased mainly due to various expenses. For other operating income (expense), an income of ¥16.1 billion was recorded, mainly due to the recognition of income from disposal of product rights and gain on sale of property, plant and equipment. As a result, Core operating profit totaled ¥450.7 billion (-¥1 billion, -0.2%), unchanged from the same period in the previous fiscal year, and Core net income increased to ¥333.6 billion (+¥15.9 billion, +5.0%) due to a decrease in income tax, and an improvement in financial income and expenses.
Reflecting the results and based on our dividend policy, Chugai plans to pay year-end dividends of ¥40 per share. As a result, the annual dividend will be ¥80 per share, and the Core dividend payout ratio is 40.9% on a five-year average basis (39.5% on a single fiscal year basis).
<R&D activities>
The company also made good progress in research and development towards achieving TOP I 2030, in both early and late stages of developments. For in-house projects that will drive mid to long-term growth, in early-stage projects, ALPS12 and ROSE12 entered in clinical development for solid tumors, and SAIL66 too for CLDN6 positive solid tumors, which Chugai’s proprietary antibody engineering technologies have been applied. For mid-size molecules, in addition to the development of the first project, LUNA18, which was confirmed absorption (blood transfer) after oral administration, Chugai is also working on approximately 30 of follow-on projects before entering clinical development. In late-stage development, Chugai filed for approval of crovalimab for PNH simultaneously in Japan, the United States and Europe. Alecensa demonstrated significant reduction in the risk of recurrence and death in the global phase III ALINA study as an adjuvant therapy for ALK-positive non-small cell lung cancer, and led to an application for an additional indication in Japan, the U.S., and Europe. In-house projects licensed to third parties excluding Roche also progressed steadily. Eli Lilly's development of orforglipron has yielded positive results in phase II studies in patients with type 2 diabetes and obesity, and phase III studies have been initiated. Nemolizumab, which is being developed overseas by Galderma, has met its primary endpoints in a phase III study for atopic dermatitis and prurigo nodularis and is progressing toward global launch in multiple indications. As for projects in-licensed from Roche, an application was filed for approval for the indication of retinal vein occlusion (RVO), the third indication, for the ophthalmic drug Vabysmo. In addition, Phesgo, a subcutaneous formulation that combines fixed doses of Herceptin and Perjeta, which has been used as a standard treatment for HER2-positive breast cancer, was approved and launched for the treatment of HER2-positive breast cancer and colorectal cancer. In 2023, to further accelerate our drug discovery engine through open innovation, we established the Chugai Venture Fund, LLC (Chugai Venture Fund, CVF), a corporate venture capital fund, in the Boston area of the US, and has started full-scale operation of investment activities from this year.
<Full year forecast for 2024>
In 2024, Core revenues, Core operating profit, and Core net income are expected to be ¥1,070.0 billion (-¥41.4 billion, -3.7%), ¥460.0 billion (+¥9.3 billion, +2.1%), and ¥335.5 billion (+¥1.9 billion, +0.6%), resulting in decrease in revenues and increase in profits. Sales are expected to decrease in Japan and increase overseas, totaling ¥922.0 billion (-¥52.5 billion, -5.4%). Domestic sales are expected to decrease to ¥454.9 billion (-¥103.1 billion, -18.5%) due to the reduction in the supply of Ronapreve to the government, the impact of the NHI drug price revision and the market penetration of generics, despite higher volumes of new products Phesgo, Vabysmo and mainstay products. Overseas sales are expected to increase to ¥467.1 billion (+¥50.6 billion, +12.1%) due to strong growth in sales of Hemlibra, including the impact of the weaker yen, despite decrease in Actemra®. Other revenues are expected to reach ¥148.0 billion (+¥11.1 billion, +8.1%). Royalty and profit-sharing income are forecasted to increase to ¥134.4 billion (+5.4%), due to an increase in income related to Hemlibra in addition to an increase in one-time income, despite a decrease in income related to Actemra.
For the fiscal year 2024, Chugai expects annual dividends per share of ¥82 with a Core dividend payout ratio of 40.2% on a five-year average basis (40.2% on a single fiscal year basis).
【2023 full year results】
| Billion JPY | 2023 | 2022 | % Change |
|---|---|---|---|
| Core results | |||
| Revenue | 1,111.4 | 1,167.8 | -4.8% |
| Sales | 974.5 | 1,039.2 | -6.2% |
| Other revenue | 136.9 | 128.6 | +6.5% |
| Operating profit | 450.7 | 451.7 | -0.2% |
| Net income | 333.6 | 317.7 | +5.0% |
| IFRS results* | |||
| Revenue | 1,111.4 | 1,259.7 | -11.8% |
| Operating profit | 439.2 | 533.3 | -17.6% |
| Net income | 325.5 | 374.4 | -13.1% |
*IFRS results in 2022 include non-Core items, such as the income and other related items, which totaled ¥90.7 billion associated with the settlement agreement between Chugai and Alexion Pharmaceuticals, Inc., which are excluded from the Core results Chugai adopts to manage recurring business activities.
<Sales breakdown>
| Billion JPY | 2023 | 2022 | % change |
|---|---|---|---|
| Sales | 974.5 | 1,039.2 | -6.2% |
| Domestic sales | 558.0 | 654.7 | -14.8% |
| Oncology | 260.2 | 256.0 | +1.6% |
| Specialty | 297.8 | 398.6 | -25.3% |
| Overseas sales | 416.5 | 384.6 | +8.3% |
<Oncology field (Domestic) Top5-selling medicines>
| Billion JPY | 2023 | 2022 | % change |
|---|---|---|---|
| Tecentriq | 65.5 | 60.9 | +7.6% |
| Avastin | 49.8 | 67.5 | -26.2% |
| Polivy | 35.5 | 15.5 | +129.0% |
| Perjeta | 33.6 | 32.3 | +4.0% |
| Alecensa | 30.3 | 28.9 | +4.8% |
<Specialty field (Domestic) Top5-selling medicines>
| Billion JPY | 2023 | 2022 | % change |
|---|---|---|---|
| Ronapreve* | 81.2 | 203.7 | -60.1% |
| Hemlibra | 54.8 | 49.3 | +11.2% |
| Actemra | 44.3 | 42.8 | +3.5% |
| Enspryng | 23.9 | 16.7 | +43.1% |
| Vabysmo | 15.3 | 6.4 | +139.1% |
*Ronapreve has not been listed in the National Health Insurance (NHI) price list.
【2024 full year forecast】
| Billion JPY | 2024 Forecast | 2023 Actual | % Change |
|---|---|---|---|
| Core-basis | |||
| Revenues | 1,070.0 | 1,111.4 | -3.7% |
| Operating profit | 460.0 | 450.7 | +2.1% |
| Net income | 335.5 | 333.6 | +0.6% |
【Progress in R&D activities from Oct 25th, 2023 to Feb 1st, 2024】
About Core results
Chugai discloses its results on a Core basis from 2013 in conjunction with its decision to apply IFRS. Core results are the results after adjusting Non-Core items to IFRS results. Chugai’s recognition of non-recurring items may differ from that of Roche due to the difference in the scale of operations, the scope of business and other factors. Core results are used by Chugai as an internal performance indicator, for explaining the underlying business performance both internally and externally, and as the basis for payment-by-results such as a return to shareholders.
Trademarks used or mentioned in this release are protected by law.
Contact:
- For Media
- Chugai Pharmaceutical Co., Ltd.
- Media Relations Group, Corporate Communications Dept.,
- Hideki Sato
- Tel: +81-3-3273-0881
- E-mail: pr@chugai-pharm.co.jp
- For Investors
- Chugai Pharmaceutical Co., Ltd.
- Investor Relations Group, Corporate Communications Dept.,
- Takayuki Sakurai
- Tel: +81-3-3273-0554
- E-mail: ir@chugai-pharm.co.jp





